Defining the immunomodulatory effects of IMM-101: a ... › wp-content › uploads › 2018 ›...
Transcript of Defining the immunomodulatory effects of IMM-101: a ... › wp-content › uploads › 2018 ›...

Media IMM-1010
200
400
600
IFNγ
(ng/
ml) IL-12p35-/- IMM-101 DCs → WT Recipient
WT IMM-101 DCs → WT Recipient
Media IMM-1010
2
4
6
8
10
IL-1
7 (n
g/m
l)
WT IMM-101 DCs → IL-12p35-/- Recipient
WT IMM-101 DCs → WT Recipient
Medium IMM-101 anti-CD30
400
800
1200
IFNγ
(ng/
ml)
DCs → SPFDCs → GF
IMM-101 DCs → SPFIMM-101 DCs → GF
Contact: [email protected]
Defining the immunomodulatory effects of IMM-101: a promising, novel co-therapy for cancer.
Alicia A. Galdon1, James Crooks1, Sheila L. Brown1, Jaap Kampinga2, Laura Rosa Brunet3 and Andrew S. MacDonald1. 1Manchester Collaborative Centre for Inflammation Research, The University of Manchester, Manchester, UK
2Immodulon Therapeutics, London, UK 3RB Consulting Ltd, London, UK
1. Background • IMM-101 is a non-specific immunomodulator containing heat-killed,
whole cell Mycobacterium obuense (NCTC13365). • IMM-101 is proposed to induce a protective CD8+ response in
clinically relevant models of pancreatic cancer (Elia et al. 2013). • The IMAGE-1 phase II clinical trial (NCT01303172) with IMM-101
demonstrated long term survival of patients with metastatic pancreatic cancer (Dalgleish et al. 2016).
• Defining how IMM-101 exerts its immunomodulatory effects is key for advancing its use as a cancer therapy.
Elia A et al., 2013, Treatment with IMM-101 induces protective CD8+ T cell responses in clinically relevant models of pancreatic cancer. J Immunother Cancer 1: Sup 1, P215 Dalgleish et al. 2016, Randomised, open-label, phase II study of gemcitibine with and without IMM-101 for advanced pancreatic cancer. British Journal of Cancer, Vol 115. 989-796
References
2. Does IMM-101 activate DCs in vitro?
A
3. What response do IMM-101 - activated DCs induce in vivo?
5. Is there a role for commensal cross-reactivity?
6. Summary
4. Role of IL-12 in IMM-101 - activated DC IFNγ induction?
B
Figure 5 - IFNγ induction by IMM-101 DCs does not require their ability to produce IL-12, but recipient IL-12 is essential. A) WT or IL-12p35-/-GMDCs stimulated with IMM-101 were injected subcutaneously into WT mice, or B) WT GMDCs activated with IMM-101 were injected into WT or IL-12p35-/- mice. After 7d, draining LNs were harvested, and isolated lymphocytes were cultured for 72h +/- IMM-101. Cytokine secretion was measured by ELISA. *p<0.05, **p<0.01, ****p<0.0001. Data representative of 3 experiments.
Figure 4 - Adoptive transfer of IMM-101 activated DCs induces IFNγ production by a range of recipient cell types. GMDCs +/- 300µg/ml IMM-101 were injected subcutaneously into WT or IFNγ-eYFP reporter mice. After 7d, draining lymph nodes were harvested. A) Isolated lymphocytes were cultured for 72h either alone or with 300µg IMM-101 or 0.5µg αCD3, and cytokine secretion measured by ELISA. Type 2 cytokines were not induced (data not shown). B) Proportions of YFP+ cells measured by FACS were used to calculate numbers of IFNγ secreting cells. Although total numbers of IFNγ+ cells increased in IMM-101 DC recipients (bar graph), proportions of IFNγ+ cells remained unchanged (pie chart). As well as IFNγ, IL-17 secretion was also increased (not shown). *p<0.05, ***p<0.001.
DCs
IMM-‐101 DCs
Re-stimulation Condition
A
T cells only
T cells + media pulsed DCs
T cells + IMM-‐101 pulsed DCs
Re-stimulation Condition
Figure 6 - IFNγ induction by IMM-101 DCs does not require recipient pre-conditioning by commensals. GMDCs +/- 300µg/ml IMM-101 were injected subcutaneously into SPF or gnotobiotic (GF) mice. After 7d, draining LNs were harvested and isolated lymphocytes cultured for 72h +/- 300µg/ml IMM-101 or 0.5µg αCD3. Cytokine secretion was measured by ELISA. In addition to IFNγ, no significant differences were found for any other cytokine in GF animals. Data representative of 3 experiments.
• I M M - 1 0 1 t r i g g e r s d o s e -dependent activation of DCs in vitro.
• IMM-101 activated DCs induce IFNγ production by CD4+, CD8+ and γδ T cells, NK cells and NKT cells in vivo.
• IFNγ induction by IMM-101 DCs does not require their ability to produce IL-12, but is dependent on recipient IL-12 production.
• The microbiota does not play a major role in the IFNγ response induced by IMM-101 DCs.
• Future work is looking to cha rac te r i se t he i n v i vo response to IMM-101 following intradermal injection, focussing on DCs, CD4+ and CD8+ T cells, and monocytes.
Figure 2 - IMM-101 causes dose-dependent activation of DCs in vitro. GMDCs were stimulated with LPS or Pam3Csk4 at 250ng/ml, or IMM-101 at 10, 100 or 300µg/ml for 18h before cells and supernatants were analysed by A) flow cytometry and B) ELISA, respectively. Similar DC activation was also seen in murine DCs generated with FLT3-L and human moDCs. *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001. Data representative of 3 experiments.
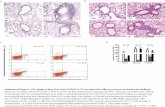
Figure 1 - DC uptake of IMM-101 in vitro. Isolated C57BL/6 bone marrow cells were cultured with GMSCF for 10d, with media changed on d3, 6 and 8. At d10, GMDCs were stimulated with 300µg/ml SYTO-9 labelled IMM-101 for 18h. Internalisation of bacteria was visualised by ImageStream.
Figure 3 - IMM-101 enhances the ability of DCs to activate OVA-specific CD4+ T cells in vitro. GMDCs were incubated for 2h +/- 300µg/ml IMM-101, then co-cultured with CFSE labelled CD4+ T cells +/- 5µg/ml OVA protein for 72h. A) The ability of IMM-101 to enhance DC induced T cell proliferation was assessed via CFSE dilution. B) DC-induced cytokine secretion was measured by ELISA. As well as IFNγ, IL-17 was induced (not shown). *p<0.05, **p<0.01, ****p<0.0001. Data representative of 2 experiments.
Media OVA0
10
20
30
IFNγ
(ng/
ml) DCs
IMM-101 DCsT cells alone
**** ****
Undivided
1 divi
sion
2 divi
sions
3 divi
sions
4 divi
sions
5 divi
sions
0
10
20
30
40
% o
f CD
4+
DCsIMM-101 DCs
**
**
*
*
*
A
B
DCs IMM-101 DCs0
1×105
2×105
3×105
Num
ber o
f YFP
+ve
cel
ls/L
N GD T cellsNK CellsNK T cells
CD4+ cellsCD8+ cells
*
***
***
***
***
***
***
Re-‐s;mula;on Condi;on
Media IMM-1010
200
400
600
IFNγ
(ng/
ml)
Media IMM-1010
200
400
600
IFNγ
(ng/
ml)
Re-‐s;mula;on Condi;on
Media IMM-1010
1
2
3
4
5
IL-1
7 (n
g/m
l)
CD86
IMM-‐101
DC
MHC-‐II CD80
CD40
TNFα
IL-‐23
CD4+ T Cell
IL-‐17 IFNγ
ProliferaCon
IL-‐6
WT Recipients IL-‐12p35-‐/-‐ Recipients
= No IFNγ response =
Strong IFNγ
response
NK cells CD4/CD8 T cells γδ T cells NK T cells
IMM-‐101 ac;vated DCs do not need to be able to produce IL-‐12 to induce IFNγ in vivo.
Recipient mice need to have resident cells able to produce IL-‐12 to induce IFNγ
in vivo.
B
A
****
Media IMM-1010
2
4
6
8
IL-1
7 (n
g/m
l)
WT IMM-101 DCs → IL-12p35-/- Recipient
WT IMM-101 DCs → WT Recipient
**
*
CD8+ T Cell
NK Cell
γδ T Cell
Medium IMM-101 anti-CD30
200
400
600
800
IFNγ
(ng/
ml)
******
Media DCsIMM-101 DCs
PBSLP
SPAM3 1010
030
00
5
10
15
TNF
(ng/
ml)
IMM-101
*
***
******
PBSLP
SPAM3 1010
030
00.00
0.05
0.10
0.15
0.20
IL-1
2p70
(ng/
ml)
IMM-101
**B
PBSLP
SPAM3 1010
030
00
100
200
300
400
IL-1
2p40
(ng/
ml)
IMM-101
***
*****