Oral hypoglycemic agents in type 2 diabetes. Type 2 diabetes is a disease of progressive β-cell...
-
Upload
beatrix-adams -
Category
Documents
-
view
226 -
download
2
Transcript of Oral hypoglycemic agents in type 2 diabetes. Type 2 diabetes is a disease of progressive β-cell...

Oral hypoglycemic agents Oral hypoglycemic agents in type 2 diabetesin type 2 diabetes

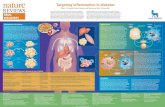
Type 2 diabetes is a disease of Type 2 diabetes is a disease of progressive progressive ββ--cellcell dysfunctiondysfunction in presence of in presence of insulin insulin resistanceresistance, leading to gradual loss of glycemic , leading to gradual loss of glycemic control. An initial loss of first phase insulin control. An initial loss of first phase insulin response contributes to elevated postprandial response contributes to elevated postprandial plasma glucose concentrations.plasma glucose concentrations.

Insulin resistance is evident in skeletal Insulin resistance is evident in skeletal muscle, liver, and adipose tissue, the major muscle, liver, and adipose tissue, the major target tissues of insulin action. target tissues of insulin action. Skeletal Skeletal musclemuscle insulin resistance leads to insulin resistance leads to post-post-prandial hyperglycemiaprandial hyperglycemia, while , while hepatichepatic insulin resistance is a causative factor in the insulin resistance is a causative factor in the subsequent development of subsequent development of fasting fasting hyperglycemia. hyperglycemia.

Sites of action of the current pharmacological Sites of action of the current pharmacological therapies for the treatmenttherapies for the treatment of type 2 diabetesof type 2 diabetes..

sulfonylureassulfonylureas
Insulin secretagogues stimulate insulin Insulin secretagogues stimulate insulin secretion by interacting with the ATP-secretion by interacting with the ATP-sensitive potassium channel on the beta sensitive potassium channel on the beta cell cell . . In general, sulfonylureas increase In general, sulfonylureas increase insulin acutely and thus should be taken insulin acutely and thus should be taken shortly before a meal; with chronic therapy, shortly before a meal; with chronic therapy, though, the insulin release is more though, the insulin release is more sustained. sustained.

sulfonylureassulfonylureas
At maximum doses, first-generation At maximum doses, first-generation sulfonylureas are similar in potency to sulfonylureas are similar in potency to second-generation agents but have a longer second-generation agents but have a longer half-life, a greater incidence of half-life, a greater incidence of hypoglycemia, and more frequent drug hypoglycemia, and more frequent drug interactions. interactions.

sulfonylureassulfonylureas
An advantage to a An advantage to a more rapid onsetmore rapid onset of of action is better coverage of the postprandial action is better coverage of the postprandial glucose rise, but theglucose rise, but the shorter half-life shorter half-life of such of such agents requires more than once-a-day agents requires more than once-a-day
dosingdosing

sulfonylureassulfonylureas The The most responsivemost responsive patients are those who patients are those who
exhibit exhibit mild-to-moderate fasting mild-to-moderate fasting hyperglycemiahyperglycemia (<200-240 mg/dl; along with (<200-240 mg/dl; along with adequate residualadequate residualββ -cell function. These -cell function. These drugs are most effective in individuals with drugs are most effective in individuals with type 2 DM of relatively recent onset type 2 DM of relatively recent onset (<5 (<5 yearsyears), who have residual endogenous ), who have residual endogenous insulin production.insulin production.

sulfonylureassulfonylureas The The higher the fasting glucosehigher the fasting glucose level, the level, the
greater the effectgreater the effect will be. In patients with a will be. In patients with a pre-treatment glucose level of approximately pre-treatment glucose level of approximately 200 mg/dl, sulfonylureas typically will 200 mg/dl, sulfonylureas typically will reduce glucose by 60-70 mg/dl and HbA1C reduce glucose by 60-70 mg/dl and HbA1C by 1.5-2%by 1.5-2%..

Relationship between baseline HbA1C and reduction in Relationship between baseline HbA1C and reduction in HbA1C from baseline achieved following treatment HbA1C from baseline achieved following treatment
with oral antihyperglycemic medication.with oral antihyperglycemic medication.

sulfonylureassulfonylureas Thus, expectations for the overall magnitude Thus, expectations for the overall magnitude
of effect from a given agent might be overly of effect from a given agent might be overly optimistic when treating patients whose optimistic when treating patients whose baseline HbA1c is <7.5-8.0%. baseline HbA1c is <7.5-8.0%.

sulfonylureassulfonylureas Approximately Approximately 10-20%10-20% of patients will of patients will
exhibit a exhibit a primary failures.primary failures. While these While these patients are typically those who have severe patients are typically those who have severe fasting hyperglycemia (>280 mg/dl; and fasting hyperglycemia (>280 mg/dl; and reduced fasting Cpeptide levels, these tests reduced fasting Cpeptide levels, these tests are not specific enough to help decide on are not specific enough to help decide on the usefulness of a sulfonylurea for an the usefulness of a sulfonylurea for an individual patient. In addition, treatment individual patient. In addition, treatment with sulfonylureas results in the eventual with sulfonylureas results in the eventual loss of therapeutic effectiveness loss of therapeutic effectiveness (secondary (secondary failure)failure) in the range of in the range of 3-10% per year3-10% per year..

Adverse effectsAdverse effects Mild hypoglycemic events in 2-4% Mild hypoglycemic events in 2-4% severe hypoglycemia 0.2-0.4 cases per 1000 severe hypoglycemia 0.2-0.4 cases per 1000
patient-yearspatient-years

sulfonylureassulfonylureas Sulfonylureas reduce both Sulfonylureas reduce both fastingfasting and and
postprandial glucosepostprandial glucose and should be initiated and should be initiated at low doses and increased at 1- to 2-week at low doses and increased at 1- to 2-week intervals based on SMBGintervals based on SMBG. .

meglitinidesmeglitinides The mechanism of action of meglitinides also The mechanism of action of meglitinides also
involves the binding to and involves the binding to and closure of the KATPclosure of the KATP channel resulting in membrane depolarization, an channel resulting in membrane depolarization, an influx of calcium, and insulin exocytosis . The influx of calcium, and insulin exocytosis . The kinetics of interactionkinetics of interaction of nateglinide with the KATP of nateglinide with the KATP channel are distinct compared to both repaglinide channel are distinct compared to both repaglinide and sulfonylureas, and accounts for its rapid and sulfonylureas, and accounts for its rapid insulinotropic effects.insulinotropic effects.

meglitinidesmeglitinides
Unlike sulfonylureas, Unlike sulfonylureas, meglitinidesmeglitinides stimulate stimulate first-phase insulin release in a first-phase insulin release in a glucose-glucose-sensitive mannersensitive manner, theoretically reducing the , theoretically reducing the risk of hypoglycemic events. The delivery of risk of hypoglycemic events. The delivery of insulin as an early, transient ‘burst’ at the insulin as an early, transient ‘burst’ at the initiation of a meal affords several major initiation of a meal affords several major physiological benefitsphysiological benefits

meglitinidesmeglitinides These include These include rapidly suppressing hepatic rapidly suppressing hepatic
glucose productionglucose production and reducing the and reducing the stimulus for additional insulin that would be stimulus for additional insulin that would be required subsequently to dispose of a larger required subsequently to dispose of a larger glucose load. Thus, the rapid onset/short glucose load. Thus, the rapid onset/short duration stimulation of insulin release by duration stimulation of insulin release by meglitinides should enhance meglitinides should enhance control of control of prandial hyperglycemia,prandial hyperglycemia, while reducing the while reducing the risk for post-absorptive hypoglycemia and risk for post-absorptive hypoglycemia and limiting exposure to hyperinsulinemia.limiting exposure to hyperinsulinemia.

meglitinidesmeglitinides Repaglinide is approximately 5 times more Repaglinide is approximately 5 times more
potent than glyburide in stimulating insulin potent than glyburide in stimulating insulin secretion. Unlike glyburide, repaglinide secretion. Unlike glyburide, repaglinide does not stimulate insulin secretion in vitro does not stimulate insulin secretion in vitro in the absence of glucose. Rather, it in the absence of glucose. Rather, it enhances glucose-stimulated insulin enhances glucose-stimulated insulin secretion especially at 180mg/dl glucose.secretion especially at 180mg/dl glucose.

meglitinidesmeglitinides As a result of more rapid absorption, it produces As a result of more rapid absorption, it produces
a generally faster and briefer stimulus to insulin a generally faster and briefer stimulus to insulin secretion. As a result, it is generally taken with secretion. As a result, it is generally taken with each meal and provides better postprandial each meal and provides better postprandial control and generally less hypoglycemia and control and generally less hypoglycemia and weight gain than glyburide. Repaglinide does weight gain than glyburide. Repaglinide does seem to have a long residence time on the seem to have a long residence time on the sulfonylurea receptor and a prolonged effect on sulfonylurea receptor and a prolonged effect on fasting glucose, even though its pharmacologic fasting glucose, even though its pharmacologic half-life is quite short. half-life is quite short.

meglitinidesmeglitinides The onset of action of netaglinide is similar to The onset of action of netaglinide is similar to
that of glyburide but three-fold more rapid than that of glyburide but three-fold more rapid than that of repaglinide . When netaglinide is removed that of repaglinide . When netaglinide is removed from the KATP channel, its effect is reversed from the KATP channel, its effect is reversed twice as quickly as glyburide and five times more twice as quickly as glyburide and five times more quickly than repaglinide. Thus, netaglinide quickly than repaglinide. Thus, netaglinide initiates a more rapid release of insulin that is initiates a more rapid release of insulin that is shorter in duration compared to shorter in duration compared to repaglinide ,despite having an in vivo repaglinide ,despite having an in vivo pharmacokinetic profile that is similar.pharmacokinetic profile that is similar.

ThiazolidinedionesThiazolidinediones These drugs bind to the These drugs bind to the PPAR-PPAR-δδnuclear nuclear
receptorreceptor. The PPAR- . The PPAR- δδ receptor is found at receptor is found at highest levels in adipocytes but is expressed highest levels in adipocytes but is expressed at lower levels in many other tissues. at lower levels in many other tissues. Agonists of this receptor regulate a large Agonists of this receptor regulate a large number of genes, number of genes, promote adipocyte promote adipocyte differentiationdifferentiation, reduce hepatic fat , reduce hepatic fat accumulation, and appear to accumulation, and appear to reduce insulin reduce insulin resistanceresistance indirectly by enhancing fatty acid indirectly by enhancing fatty acid storage and possibly by increasing storage and possibly by increasing adiponectin levelsadiponectin levels

ThiazolidinedionesThiazolidinediones It does not appear that rosiglitazone and It does not appear that rosiglitazone and
pioglitazone improve insulin sensitivity and pioglitazone improve insulin sensitivity and glucose disposal by direct effects on either liver glucose disposal by direct effects on either liver or muscle. PPARor muscle. PPARδδ is expressed chiefly in adipose is expressed chiefly in adipose tissue, and its expression in liver and skeletal tissue, and its expression in liver and skeletal muscle is low .Thus, it is more likely that the muscle is low .Thus, it is more likely that the primary effects of these drugs are on adipose primary effects of these drugs are on adipose tissue, followed by secondary benefits on other tissue, followed by secondary benefits on other target tissues of insulin.target tissues of insulin.

ThiazolidinedionesThiazolidinediones Thiazolidinediones promote a redistribution of fat Thiazolidinediones promote a redistribution of fat
from central to peripheral locations. Circulating from central to peripheral locations. Circulating insulin levels decrease with use of the insulin levels decrease with use of the thiazolidinediones, indicating a reduction in insulin thiazolidinediones, indicating a reduction in insulin resistanceresistance..

ThiazolidinedionesThiazolidinediones
Each drug as monotherapy, results in a Each drug as monotherapy, results in a significant reduction in fasting plasma glucose significant reduction in fasting plasma glucose by 60-80 mg/dl and in HbA1c by 1.4-2.6% . In by 60-80 mg/dl and in HbA1c by 1.4-2.6% . In addition, pioglitazone is approved for use in addition, pioglitazone is approved for use in combination with insulin, metformin, or a combination with insulin, metformin, or a sulfonylurea, and rosiglitazone is approved for sulfonylurea, and rosiglitazone is approved for use in combination with metformin or a use in combination with metformin or a sulfonylurea. sulfonylurea.

ThiazolidinedionesThiazolidinediones Unlike other existing anti-diabetic Unlike other existing anti-diabetic
medications that possess a very rapid onset medications that possess a very rapid onset of activity, TZD exhibit a characteristic delay of activity, TZD exhibit a characteristic delay from 4-12 weeks in the onset of their from 4-12 weeks in the onset of their therapeutic benefits. therapeutic benefits.

ThiazolidinedionesThiazolidinediones Patients treated with 15, 30, or 45 mg (once daily) Patients treated with 15, 30, or 45 mg (once daily)
pioglitazone had significant mean decreases in HbA1c pioglitazone had significant mean decreases in HbA1c (range -1.00 to -1.60%) and fasting plasma glucose (-39.1 (range -1.00 to -1.60%) and fasting plasma glucose (-39.1 to -65.3 mg/dl). The decreases in fasting plasma glucose to -65.3 mg/dl). The decreases in fasting plasma glucose were observed as early as the second week of therapy; were observed as early as the second week of therapy; maximal decreases occurred after 10-14 weeks maximal decreases occurred after 10-14 weeks ..

Due to their mechanism of action, the Due to their mechanism of action, the risk risk of hypoglycemiaof hypoglycemia with rosiglitazone or with rosiglitazone or pioglitazone monotherapy pioglitazone monotherapy is lowis low. Mild to . Mild to moderate hypoglycemia has been reported moderate hypoglycemia has been reported during combination therapy with during combination therapy with sulfonylureas or insulin .sulfonylureas or insulin .

Side effetsSide effets weight gain (2–3 kg)weight gain (2–3 kg) small reduction in the hematocrit small reduction in the hematocrit mild increase in plasma volume. mild increase in plasma volume. Peripheral edema and CHF is more Peripheral edema and CHF is more
common in individuals also treated with common in individuals also treated with insulin. insulin.
rosiglitazone is associated with an rosiglitazone is associated with an increased risk of myocardial infarction.increased risk of myocardial infarction.
worsening of diabetic macular edema. worsening of diabetic macular edema. increased risk of fractures has been increased risk of fractures has been
noted in women taking these agents. noted in women taking these agents.

contraindicationscontraindications liver disease liver disease CHF (class III or IV). CHF (class III or IV).

ThiazolidinedionesThiazolidinediones
Although rosiglitazone and pioglitazone do Although rosiglitazone and pioglitazone do not appear to induce the liver abnormalities, not appear to induce the liver abnormalities, the FDA recommends measurement of liver the FDA recommends measurement of liver function tests prior to initiating therapy with function tests prior to initiating therapy with a thiazolidinedione and at regular intervals a thiazolidinedione and at regular intervals (every 2 months for the first year and then (every 2 months for the first year and then periodically).periodically).

ThiazolidinedionesThiazolidinediones With regard to edema, with appropriate With regard to edema, with appropriate
caution almost no one should need to caution almost no one should need to withdraw from therapy as a result of fluid withdraw from therapy as a result of fluid retention. The patients most likely to retention. The patients most likely to experience edema are those treated with experience edema are those treated with insulin and those with preexisting edemainsulin and those with preexisting edema. . Thus, Thus, womenwomen, , overweight patientsoverweight patients, and , and those with those with diastolic dysfunctiondiastolic dysfunction or or renal renal insufficiencyinsufficiency are at greatest risk are at greatest risk

ThiazolidinedionesThiazolidinediones In the previously edematous patient and in In the previously edematous patient and in
patients treated with insulin, it is prudent to patients treated with insulin, it is prudent to initiate therapy with the lowest available initiate therapy with the lowest available dose of glitazone. In 1 to 3 months, if the dose of glitazone. In 1 to 3 months, if the glycemic response has been inadequate and glycemic response has been inadequate and significant edema has not developed, significant edema has not developed, consider increasing the dose of glitazone consider increasing the dose of glitazone further with continued expectant home further with continued expectant home evaluation for edema.evaluation for edema.

BiguanidesBiguanides
Metformin Metformin is representative of this class of is representative of this class of agents. It agents. It reduces hepatic glucose reduces hepatic glucose productionproduction through an undefined through an undefined mechanism and improves peripheral glucose mechanism and improves peripheral glucose utilization slightlyutilization slightly . .

MetforminMetformin metformin metformin reduces fasting plasma insulinreduces fasting plasma insulin and and
improve insulin sensitivity. While it is possible that improve insulin sensitivity. While it is possible that the beneficial effect of metformin on insulin the beneficial effect of metformin on insulin sensitivity is mediated directly, a more likely sensitivity is mediated directly, a more likely explanation is that it is secondary to a reduction in explanation is that it is secondary to a reduction in hyperglycemia, triglycerides, and free fatty acids.hyperglycemia, triglycerides, and free fatty acids.

EfficacyEfficacy
A large number of well-controlled clinical A large number of well-controlled clinical studies have established that metformin studies have established that metformin monotherapy consistently reduces FBS by monotherapy consistently reduces FBS by 60-70 mg/dl and HbA1c by 1.5-2.0% . Similar 60-70 mg/dl and HbA1c by 1.5-2.0% . Similar to the sulfonylurea treatment, the overall to the sulfonylurea treatment, the overall magnitude of response to metformin is magnitude of response to metformin is directly related to the starting FBS directly related to the starting FBS concentration.concentration.

MetforminMetformin Unlike sulfonylurea treatment, metformin Unlike sulfonylurea treatment, metformin
monotherapy is not associated with weight monotherapy is not associated with weight gain and even promotes a modest weight gain and even promotes a modest weight loss. loss.

MetforminMetformin
The initial starting dose of 500 mg once or The initial starting dose of 500 mg once or twice a day can be increased to 1000 mg bidtwice a day can be increased to 1000 mg bid

MetforminMetformin Because of its relatively slow onset of Because of its relatively slow onset of
action and gastrointestinal symptoms action and gastrointestinal symptoms with higher doses, the dose should be with higher doses, the dose should be escalated every 2–3 weeks based on escalated every 2–3 weeks based on
SMBG measurementsSMBG measurements..

Effect ofEffect of metformin on fasting plasma metformin on fasting plasma glucose when given as add-on therapyglucose when given as add-on therapy to glyburideto glyburide..

MetforminMetformin When added to a sulfonylurea, the effects of both agents When added to a sulfonylurea, the effects of both agents
are additive, consistent with their different mechanisms of are additive, consistent with their different mechanisms of action. Interestingly, in patients that no longer responded action. Interestingly, in patients that no longer responded to sulfonylurea treatment (secondary failures) and were to sulfonylurea treatment (secondary failures) and were removed from treatment, addition of metformin had removed from treatment, addition of metformin had minimal effects . Thus, in these patients, sulfonylurea minimal effects . Thus, in these patients, sulfonylurea treatment was still eliciting an effect, emphasizing the treatment was still eliciting an effect, emphasizing the need to continue treatment with both agents.need to continue treatment with both agents.

Side Effects and Side Effects and ContraindicationsContraindications
The The most commonmost common are are GIGI disturbances disturbances (abdominal discomfort, diarrhea), in 20-(abdominal discomfort, diarrhea), in 20-30% of patients. These effects are 30% of patients. These effects are generally transient, and can be minimized generally transient, and can be minimized or avoided by careful dose titration.or avoided by careful dose titration.

The incidence of The incidence of lactic acidosis is lactic acidosis is rarerare and occurs with a frequency of and occurs with a frequency of 3 cases per 100,000 patient-years.3 cases per 100,000 patient-years.

The risk of lactic acidosis can be minimized The risk of lactic acidosis can be minimized when the following are consideredwhen the following are considered::
1.Withhold in conditions predisposing 1.Withhold in conditions predisposing to renal insufficiency and/or hypoxiato renal insufficiency and/or hypoxia
aa. CV collapse. CV collapse b. Acute MI or acute CHFb. Acute MI or acute CHF c. Severe infectionc. Severe infection d. Use of iodinated contrast materiald. Use of iodinated contrast material e. Major surgical procedurese. Major surgical procedures

2. 2. Metformin should not be Metformin should not be prescribed for patients with:prescribed for patients with:
aa. Renal dysfunction [e.g. Scr >1.5 mg/dl . Renal dysfunction [e.g. Scr >1.5 mg/dl (males), >1.4 mg/dl (females) or abnormal CrCl(males), >1.4 mg/dl (females) or abnormal CrCl
b. Liver dysfunctionb. Liver dysfunction c. History of alcohol abuse/binge drinkingc. History of alcohol abuse/binge drinking d. Acute or chronic metabolic acidosisd. Acute or chronic metabolic acidosis

MetforminMetformin Metformin should beMetformin should be discontinued discontinued in in
patients who are patients who are seriously illseriously ill, in patients , in patients who are who are NPONPO, and in those receiving , and in those receiving radiographic contrastradiographic contrast material. material.

αα-GLUCOSIDASE INHIBITORS-GLUCOSIDASE INHIBITORS
They bind competitively to the They bind competitively to the oligosaccharide binding site of the oligosaccharide binding site of the αα--Glucosidase enzymes, thereby preventing Glucosidase enzymes, thereby preventing enzymatic hydrolysis. Acarbose binding enzymatic hydrolysis. Acarbose binding affinity for the affinity for the αα-Glucosidase enzymes is: -Glucosidase enzymes is: glycoamylase > sucrase > maltase> glycoamylase > sucrase > maltase> dextranase . Acarbose has little affinity for dextranase . Acarbose has little affinity for isomaltase and no affinity for the isomaltase and no affinity for the ββ - -glucosidase enzymes, such as lactaseglucosidase enzymes, such as lactase

αα-Glucosidase inhibitors-Glucosidase inhibitors
acarbose and miglitol reduce postprandial acarbose and miglitol reduce postprandial hyperglycemia by delaying glucose hyperglycemia by delaying glucose absorption; they do not affect glucose absorption; they do not affect glucose utilization or insulin secretionutilization or insulin secretion

EfficacyEfficacy
Clinical trials conducted to date have Clinical trials conducted to date have established that the antihyperglycemic established that the antihyperglycemic effectiveness of acarbose is less than 50% effectiveness of acarbose is less than 50% than that of either sulfonylureas or than that of either sulfonylureas or metformin. When used as monotherapy, metformin. When used as monotherapy, acarbose primarily affects post-prandial acarbose primarily affects post-prandial glucose levels, which is reduced by 40-50 glucose levels, which is reduced by 40-50 mg/dl after mealmg/dl after meal

Side EffectsSide Effects
The major side effects are GI disturbances. The major side effects are GI disturbances. in in 25-30% 25-30% of patients, ( flatulence, diarrhea, of patients, ( flatulence, diarrhea, bloating, and abdominal discomfort.)bloating, and abdominal discomfort.)
These side effects can often be minimized by These side effects can often be minimized by careful dose titration, and sometimes careful dose titration, and sometimes diminish with time.diminish with time.
Asymptomatic and reversible; dose Asymptomatic and reversible; dose dependent hepatotoxicity, serum ALT levels dependent hepatotoxicity, serum ALT levels require monitoring for patients receiving high require monitoring for patients receiving high doses (>200 mg three times daily) doses (>200 mg three times daily)
Hypoglycemia does not occur as Hypoglycemia does not occur as monotherapymonotherapy. .

contraindicationscontraindications
inflammatory bowel diseaseinflammatory bowel disease cirrhosiscirrhosis elevated plasma creatinineelevated plasma creatinine gastroparesisgastroparesis Simultaneous treatment with resins and Simultaneous treatment with resins and
antacidsantacids..

αα-Glucosidase inhibitors-Glucosidase inhibitors
Therapy should be initiated at a low dose Therapy should be initiated at a low dose (25 mg of acarbose) with the evening meal (25 mg of acarbose) with the evening meal and may be increased to a maximal dose and may be increased to a maximal dose over weeks to months (50–100 mg acarbose over weeks to months (50–100 mg acarbose with each meal). with each meal).

αα-Glucosidase inhibitors-Glucosidase inhibitors If If hypoglycemia hypoglycemia occurs while a patient is occurs while a patient is
taking an taking an αα-glucosidase inhibitor -glucosidase inhibitor simultaneously with a sulfonylurea, insulin simultaneously with a sulfonylurea, insulin or a meglitinide, the recommended action is or a meglitinide, the recommended action is oral administration of oral administration of pure glucose, pure glucose, dextrose or milkdextrose or milk..

The Incretin EffectThe Incretin Effect
The incretin effect, defined by a significantly The incretin effect, defined by a significantly greater insulingreater insulin stimulatory effect evoked stimulatory effect evoked after an oral glucose loadafter an oral glucose load than that evoked than that evoked from an intravenous glucose infusion when from an intravenous glucose infusion when plasma glucose concentrations are matched, plasma glucose concentrations are matched, was first described in the 1960’s.was first described in the 1960’s.


Incretin MimeticsIncretin Mimetics
Incretin mimetics are compounds that mimic several of the Incretin mimetics are compounds that mimic several of the physiologic effects of GLP-1:physiologic effects of GLP-1:
enhancement of glucose-dependent insulin secretion, enhancement of glucose-dependent insulin secretion, suppression of postprandial glucagon secretion, suppression of postprandial glucagon secretion, slowing of gastric emptying, slowing of gastric emptying, reduction of food intake and body weight in human reduction of food intake and body weight in human
studies.studies. Incretin mimetics have also been shown to promote Incretin mimetics have also been shown to promote ββ-cell -cell
proliferation and neogenesis in cell lines and animal proliferation and neogenesis in cell lines and animal models.models.

Furthermore,Furthermore, GLP-1 GLP-1 also has direct effects on the also has direct effects on the ββ-cells, -cells, as shown in studies done in animal models and cell lines, as shown in studies done in animal models and cell lines, promoting cell proliferationpromoting cell proliferation and neogenesis, while and neogenesis, while preventing preventing ββ -cell apoptosis -cell apoptosis .Additionally, GLP-1 can .Additionally, GLP-1 can promote transformation of non insulin-producing promote transformation of non insulin-producing pancreatic cells into cells capable of synthesizing and pancreatic cells into cells capable of synthesizing and secreting insulin.secreting insulin.

The characteristic The characteristic ββ -cell dysfunction, seen in -cell dysfunction, seen in patients with type 2 diabetes ,most likely develops patients with type 2 diabetes ,most likely develops years before the manifestation of overt years before the manifestation of overt hyperglycemia,and it has been suggested that at the hyperglycemia,and it has been suggested that at the time of diagnosis of disease -cell mass and secretory time of diagnosis of disease -cell mass and secretory function may have declined by as much as 50%.function may have declined by as much as 50%.

The pathological The pathological loss of loss of ββ -cell function -cell function may be the result of a number of factors may be the result of a number of factors including including ββ -cell secretory defects, -cell secretory defects, glucotoxicityglucotoxicity due to hyperglycemia, due to hyperglycemia, lipotoxicitylipotoxicity due to dyslipidemia, and due to dyslipidemia, and possibly abnormalities in secretion of, or possibly abnormalities in secretion of, or response to, response to, incretin hormonesincretin hormones..

Patients with type 2 diabetes have a Patients with type 2 diabetes have a significant reduction of the incretin effect, significant reduction of the incretin effect, implying that these patients either have implying that these patients either have decreased concentration of the incretin decreased concentration of the incretin hormones, or a resistance to their effects. hormones, or a resistance to their effects.

GLP-1 concentrations are reduced in GLP-1 concentrations are reduced in patients with type 2 diabetes in response to patients with type 2 diabetes in response to a meal, while GIP concentrations are either a meal, while GIP concentrations are either normal or increased, suggesting a normal or increased, suggesting a resistance to the actions of GIP thus making resistance to the actions of GIP thus making GLP-1 a more logical target for therapeutic GLP-1 a more logical target for therapeutic intervention.intervention.

IncretinsIncretins "Incretins" amplify glucose-stimulated "Incretins" amplify glucose-stimulated
insulin secretion. Agents that either act as a insulin secretion. Agents that either act as a GLP-1 agonist or enhance endogenous GLP-GLP-1 agonist or enhance endogenous GLP-1 activity have become available and are 1 activity have become available and are under development. Exenatide, a synthetic under development. Exenatide, a synthetic version of a peptide originally found in the version of a peptide originally found in the saliva of the Gila monster (exendin-4), is an saliva of the Gila monster (exendin-4), is an analogue of GLP-1 analogue of GLP-1

Unlike native GLP-1 which has a half-life of Unlike native GLP-1 which has a half-life of <2 min, differences in the exenatide amino <2 min, differences in the exenatide amino acid sequence render it resistant to the acid sequence render it resistant to the enzyme that degrades GLP-1 (dipeptidyl enzyme that degrades GLP-1 (dipeptidyl peptidase IV,). Thus, exenatide has peptidase IV,). Thus, exenatide has prolonged GLP-1-like action by binding to prolonged GLP-1-like action by binding to GLP-1 receptors found in islets, the GLP-1 receptors found in islets, the gastrointestinal tract, and the brain. gastrointestinal tract, and the brain.

The A1C reductions with exenatide are The A1C reductions with exenatide are modest compared to those with some oral modest compared to those with some oral agents. Exenatide is approved only for use agents. Exenatide is approved only for use as adjunct or combination therapy with as adjunct or combination therapy with metformin or sulfonylureas. Exenatide metformin or sulfonylureas. Exenatide should not be used in patients taking should not be used in patients taking insulin. insulin.

The major side effects are nausea, vomiting, The major side effects are nausea, vomiting, and diarrhea; some patients taking insulin and diarrhea; some patients taking insulin secretagogues may require a reduction in secretagogues may require a reduction in those agents to prevent hypoglycemia. those agents to prevent hypoglycemia. Because it slows gastric emptying and may Because it slows gastric emptying and may influence the absorption of other drugs, the influence the absorption of other drugs, the timing of administration should be timing of administration should be coordinatedcoordinated. .

Patients with type 2 diabetes exhibit Patients with type 2 diabetes exhibit reduced levels of active GLP-1 (amino acids reduced levels of active GLP-1 (amino acids 7-36) along with an impaired GLP-1 7-36) along with an impaired GLP-1 response to a glucose load, and parenteral response to a glucose load, and parenteral administration of GLP-1 has been shown to administration of GLP-1 has been shown to reduce fasting and post-prandial glycemia in reduce fasting and post-prandial glycemia in patients with type 2 diabetes.patients with type 2 diabetes.

VildagliptinVildagliptin
A 2nd generation DPP-IV inhibitor, A 2nd generation DPP-IV inhibitor, vildagliptin is a selective and orally-effective vildagliptin is a selective and orally-effective inhibitor of DPP-4 . Vildagliptin has been inhibitor of DPP-4 . Vildagliptin has been found to increase plasma levels of intact found to increase plasma levels of intact (active) GLP-1, to suppress meal-stimulated (active) GLP-1, to suppress meal-stimulated glucagon, and to improve glucose tolerance glucagon, and to improve glucose tolerance during a standardized meal test.during a standardized meal test.

sitagliptinsitagliptin These agents promote insulin secretion in the These agents promote insulin secretion in the
absence of hypoglycemia or weight gain, and absence of hypoglycemia or weight gain, and appear to have a preferential effect on appear to have a preferential effect on postprandial blood glucose.postprandial blood glucose. The FDA has The FDA has approved the first DPP-IV inhibitor, sitagliptin, approved the first DPP-IV inhibitor, sitagliptin, for use with diet and exercise,metformine and for use with diet and exercise,metformine and TZD to improve glycemic control in adult TZD to improve glycemic control in adult individuals with type 2 DM. individuals with type 2 DM.

sitagliptinsitagliptin Sitagliptin is administered at a dose of 100 Sitagliptin is administered at a dose of 100
mg orally once daily. Reduced doses should mg orally once daily. Reduced doses should be given to patients with moderate (GFR < be given to patients with moderate (GFR < 30–50 mL/min, 50 mg once daily) or severe 30–50 mL/min, 50 mg once daily) or severe (GFR < 30 mL/min, 25 mg once daily) renal (GFR < 30 mL/min, 25 mg once daily) renal insufficiency. Renal function should be insufficiency. Renal function should be assessed prior to initiation of sitagliptin assessed prior to initiation of sitagliptin therapy and periodically thereafter. therapy and periodically thereafter.

Clinical Efficacy of Pharmacological Therapies to Treat Clinical Efficacy of Pharmacological Therapies to Treat Type 2Type 2
Diabetes when Used as MonotherapyDiabetes when Used as Monotherapy

assuming a similar degree of glycemic assuming a similar degree of glycemic improvement, no clinical advantage to one improvement, no clinical advantage to one class of drugs has been demonstrated, and class of drugs has been demonstrated, and any therapy that improves glycemic control any therapy that improves glycemic control is likely beneficial is likely beneficial ..

Emphasis should be placed on reaching Emphasis should be placed on reaching lipid, blood pressure, and glycemic targets lipid, blood pressure, and glycemic targets rather than the type of therapy needed to rather than the type of therapy needed to reach those goals .reach those goals .