Chin. Ann. Math. Ser. B Chinese Annalsof Mathematics,SeriesB
Controll ed multiple growth factor d elivery from bone ... · Michigan, Ann A ve Research Cente...
Transcript of Controll ed multiple growth factor d elivery from bone ... · Michigan, Ann A ve Research Cente...

Ssoarebrethinpfaarem(mgcrebinlugth5bsmgMimainS(mispsccwAEmrethinfedccRβrewwrhdDgdhimsSindp
Statement of Purpimple mechanism
osteogenic growthangiogenesis playselease of an angi
bone formation. Helevant approachehat mineral coatincorporation and
phosphate (β-TCPfabrication (SFF) approach. Mineral ecombinant huma
modular, mineral-mBMP2) solution
growth factor. Theculture medium anelease of both gro
biological activityntramuscular sheeumborum and coll
growth factor, rhVhe combination o
50μg mBMP2. Thblood vessel ingrohowed an increa
manner in groups rgrowth factors. Methods: β-TCP smage-based desig
achieve scaffolds nterconnected por
Scaffolds were incmSBF) for periods a solution simi
plasma. The minecanning electron m
conducted using 12
carboxyfluoresceinwere sequentially After binding, β-TEagle’s medium (Dmedia were collecteleased growth fache released mediuntensity for mBM
female sheep weredelivering differencombination from consisted of mineraResults: Our resultβ-TCP scaffolds (esults in binding o
we showed sustainwith VEGF releashVEGF in vivo in
dose dependent maDiscussion: In thisgrowth factor releadouble dipping prohighly adaptable camplanted in vivo, caffolds.
Significance: This n which a controll
device as a thin surphysical properties
Controlle
pose: The objecti for release of two
h factor, from a ss an important roiogenic and osteo
However, there remes for dual growthings could be urelease of multip
P) scaffolds weretechniques and mcoated β-TCP sc
n vascular endothbinding version
ns for one hour eace dual release profind simulated bodyowth factors for ovy of the growth p model. Scaffoldlected at 2 and 4 w
VEGF, were assessof the different dohe samples collectowth using vonWiase in blood vessreleasing either rh
scaffolds were fabgn and 3D printi
with controlled res with a 40% cubated at 37 ˚C
ds of 7 days underilar in ionic compral formed on themicroscopy (SEM
25I-labeled rhVEGFn labeled mBMP2
dipped in solutioTCP granules wereDMEM) at 37oC. ted and replaced ctor was determine
um for rhVEGF reMP2. For our in ve used. The experint dosages of rhVE
mineral coated βal coated scaffoldsts demonstrate tha(Figure 1), and tof both rhVEGF aned dual release osing more rapidly nduced an increaseanner (Figure 4). s study, we develoase of an angiogeniocess. The mineral arrier to bind and rrhVEGF increased
approach demonsable biologics carrrface coating, with of the device. Thi
ed multiple gr1Suarez-Gon
3AO Foun
ives of this study o growth factors, scaffold material. ole in bone regenogenic growth facmains a need for h factor release.
used as a platforple growth factors fabricated using
mineral coated usaffolds were sequ
helial growth factoof bone morpho
ch to allow for incfile was characterizy fluid (SBF), and ver more than 60
factors was chads were implanted weeks. Different dsed (0.5, 1, 5, andosages of rhVEGted at 2 weeks willebrand factor stsel ingrowth in ahVEGF alone or th
bricated by SFF fabing techniques warchitecture: sqvolume void fr
in modified simur continuous rotatiposition and teme material was ch
M). Binding and reF (Perkin Elmer; . Mineral coated
ons of either rhVe incubated in DuAt specific time pwith fresh mediumed by measuring tlease; or by measuvivo studies, 10 Hmental group con
EGF, mBMP-2 froβ-TCP scaffolds. Ts with no growth faat we could form mthat the “double and mBMP2 (Figuof rhVEGF and m
than mBMP2. Se in the number of
ped a simple approic and osteogenic coating served as
release rhVEGF and blood vessel ingr
strates a “modular rier is integrated in
hout negatively impis type of modular
owth factor dnzalez, D; 2Diggs,
1University of2University of
ndation Collaboratidarilis.
were to develop aan angiogenic andIt is known that
neration, and thatctors can enhance
simple, clinicallyWe hypothesized
rm for controlleds. Beta tricalciumg solid free formsing a biomimeticuentially dipped inor (rhVEGF) and agenetic protein 2orporation of eachzed in vitro in celshowed sustaineddays. The in vivoaracterized in anin the longissimus
dosages of a singled 10μg) as well as
GF combined withwere evaluated fortaining (vWF) anda dose dependenhe combination of
brication. In shortwere employed toquare, orthogonaraction (porosity)ulated body fluidson. Modified SBF
mperature to bloodharacterized usinglease studies wereBoston, MA), andd β-TCP scaffolds
VEGF or mBMP2ulbecco’s modifiedpoints, the releasem. The amount ofthe radioactivity ofuring fluorescenceHampshire mature
nsisted of scaffoldsom mineral, or theThe control groupfactor. mineral coatings on
dipping” processure 2). In addition
mBMP2 (Figure 3)Scaffolds releasingf blood vessels in a
oach for dual factor based on a a simple and
nd mBMP2. Whenrowth in the
design” approach,nto the structural pacting the bulk r design approach
elivery from bA; 1Lee, JS; 2Holl
f Wisconsin, Madif Michigan, Ann Aive Research [email protected]
a d t t e y d d
m m c n a 2 h l d o n s e s h r d t f
t, o l . s F d g e d s . d e f f e e s e p
n s
n, , g a
n
,
may bscaffobiolog
FigureTCP scontinlike na
Figuredippinamounsignifi
FigureDMEMgrowthions is
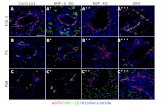
FigurearrowsvesselrhVEGPositivblood pores condit
a)
bone tissue enlister, SJ; 1,3Murphison, WI
Arbor, MI er, Davos, Switzerm
be particularly usefolds, in which theregical properties.
e 1: a) β-TCP scaffscaffolds in mSBF nuous coating. d) Tanostructure chara
e 2: a) Dual growthng of scaffolds in rnt of rhVEGF bouicantly changed du
e 3: Dual release oM. Release was suh factors was slows likely causing the
e 4: rhVEGF releas) 2 weeks after imls within sections oGF; b) 0.5μg rhVEve vWF staining isvessel. e) Quantifof the scaffold. *ption. Scale bars = 1
gineering scafhy, WL
rland
ful for design of boe is a clear need fo
ffold fabricated via
for 7 days resulteThe morphology ofacteristic of hydrox
h factor binding wrhVEGF initially, fnd to the scaffoldsuring the second d
of rhVEGF and mBustained for over 2wer compared to De slower release of
se enhanced bloodmplantation. a-d) vof implanted β-TCEGF; c) 1.0μg rhVs brown, and circufication of the nump < 0.009 relative t100μm.
b)
ffolds
one tissue engineeor optimized physi
a SFF. b-c) Incubatd in the formationf the mineral displxyapatite.
was obtained after tfollowed by mBMs after the first dip
dipping step.
BMP2 in a) SBF an months. In SBF thMEM. The re-precf growth factors.
d vessel ingrowth (vWF immunostainiCP scaffolds releasi
EGF; d) 10μg rhVular vWF staining rmber of blood vesseto the NO Growth
ering cal and
tion of β-
n of a layed a plate
the sequential
MP2. b) The ping was not
nd b) he release of cipitation of
(black ing of blood ing a) No
VEGF. represents a els within the factor
Poster No. 0656 • ORS 2012 Annual Meeting