Quantum Physics Lecture 14 - Trinity College, Dublin · Quantum Physics Lecture 14 Interatomic...
Transcript of Quantum Physics Lecture 14 - Trinity College, Dublin · Quantum Physics Lecture 14 Interatomic...

Quantum Physics Lecture 14
Interatomic bonding electron exchange and forces s & p bonds, hybridisation
Bonds & bands Semiconductors, metals, insulators
The laser
Physics of the nucleus

Bonds between atoms (cont.) Bonding
Antibonding
ψ orbital is ‘periodic’ inside atom & decaying outside
At finite separations there is a ‘barrier’ between atoms
but electrons move between atoms by tunnelling….
➞ Exponential variation of energy of interaction with separation – Interatomic forces
Expect relationship between forces and current between atoms…

Scanning Tunneling Microscope (STM)
Sharp point (tip) close (~ 1 nm) to surface; under bias electrons tunnel across the gap (barrier potential width z) Because of exponential dependence on z (factor of ~10 for 1Å change when Uo ~ 4 eV), tunnel current is very sensitive to variations in z as tip is scanned across surface. Keep current constant ⇒ z const. ⇒ tip height = image Also, exponential dependence restricts to narrow region of tunneling, giving “atomic” resolution. ⇒ Imaging atoms…
Ge on Si

Atomic Force Microscope Uses Forces (e.g. covalent)
→Atom manipulation TiO2
Eigler et al
Morita et al

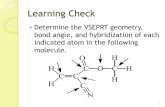
Overlapping orbitals - beyond 1s
Add the wavefunctions, noting signs!
one lobe of p-orbital is +ve the other –ve

p-orbital bonds (cont.)

Hybrid orbitals
Combine p & s states to give sp hybrid

Hybrid orbitals (cont.)
Carbon ground state
↑↓ ↑↓ ↑ ↑ 1s 2s 2p 2p 2p
C*
↑↓ ↑ ↑ ↑ ↑ 1s 2s 2p 2p 2p
Methane CH4

Bonds & Bands
A solid contains many atoms interacting, with different interaction strengths (e.g. due to separation etc.)
As a result, many bonding and anti-bonding type states, very close together. Forms ‘bands’ of energy These are the possible energy states of an electron in a solid
The band energies, and whether they are occupied by electrons is determined by the elements (i.e. Z number)

Metal or insulator or….

Semiconductors
Adding impurity (dopant) atoms with more or less electrons than semiconductor (eg. P or Ga in Si) adds electrons or holes to the bands, forming p-type and n-type.
Important for electronic devices….

The Laser
Stimulated emission produces coherent light (Bosons!) Requires population inversion & Mirror cavity
Now mostly Semiconductor lasers….

The atom nucleus
Binding energy per nucleon vs Z
Energies involved are very large…. consider via uncertainty principle!

Terms in the ‘liquid drop’ model of the nucleus Used to evaluate stability with composition - such as number of neutrons
Stability of the nucleus
Only some nuclei are stable Others decay – ‘radioactivity’

Isotopes
Same Z (number of protons) may have different number of neutrons
– isotopes of an element
Segré plot shows number of neutrons and protons for stable elements and isotopes

Radioactive decay can be spontaneous or induced