AP Chem-058 Spontaneous Processes KN PDF December 28, 2013. GraphiteUSGOV.jpg.! "File:Hex ice ......
Transcript of AP Chem-058 Spontaneous Processes KN PDF December 28, 2013. GraphiteUSGOV.jpg.! "File:Hex ice ......

Spontaneous Processes
Chemistry Essentials - 058
Nonspontaneous
Spontaneous
bozemanscience.com

Enthalpy
Processes
K > 1 K < 1
Spontaneous Not Spontaneous
Internal Energy
ΔH° < 0 ΔS° > 0 ΔH° > 0 ΔS° < 0
Products Favored
Reactants Favored
Equilibrium Constant
bozemanscience.com

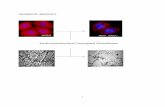
Spontaneous Processes
graphite
diamond
Nonspontaneous
Spontaneous
Thermodynamically Favored Processesbozemanscience.com

Spontaneous ProcessesEnthalpy (ΔH)
Entropy (ΔS)
bozemanscience.com

Equilibrium Constant
K=[products][reactants]
reactants products
bozemanscience.com

K=[products][reactants]100
phet.colorado.edu
Equilibrium
bozemanscience.com

K=[products][reactants]>100
phet.colorado.edu
Spontaneous
bozemanscience.com

K=[products][reactants]<100
phet.colorado.edu
Not
Spontaneous
bozemanscience.com

Spontaneous
Not spontaneous
ΔH° < 0ΔS° > 0
ΔH° > 0ΔS° < 0
bozemanscience.com

ΔH° < 0
ΔS° > 0
ΔS° < 0
ΔH° > 0
Spontaneous
Not Spontaneous
Coming soon...
Coming soon...
bozemanscience.com

To predict whether or not a process is spontaneous using ΔH° and ΔS°.
!
Did you learn?
SpontaneousΔH° < 0ΔS° > 0
bozemanscience.com

bozemanscience.com
File:Brillanten.jpg." Wikipedia, the Free Encyclopedia, December 18, 2013. http://en.wikipedia.org/w/index.php?title=File:Brillanten.jpg&oldid=404322469.!"File:GraphiteUSGOV.jpg." Wikipedia, the Free Encyclopedia. Accessed December 28, 2013. http://en.wikipedia.org/wiki/File:GraphiteUSGOV.jpg.!"File:Hex ice.GIF." Wikipedia, the Free Encyclopedia. Accessed December 29, 2013. http://en.wikipedia.org/wiki/File:Hex_ice.GIF.!"File:Josiah Willard Gibbs -from MMS-.jpg." Wikipedia, the Free Encyclopedia. Accessed December 29, 2013. http://en.wikipedia.org/wiki/File:Josiah_Willard_Gibbs_-from_MMS-.jpg.!Gkai. English: Erlenmeyer Flask, May 23, 2010. Own work. http://commons.wikimedia.org/wiki/File:Erlenmeyer-flask.svg.!Haacken, User: Herbert. English: Instant Cold Pack, March 15, 2012. Own work: Herbert Haacken. http://commons.wikimedia.org/wiki/File:2012-03-15_Ruck-Zuck-Pack_K%C3%A4lte_Katalog_Small.gif.!"Reversible Reactions." PhET. Accessed December 28, 2013. http://phet.colorado.edu/en/simulation/reversible-reactions.!Splettstoesser, Thomas. Hydrogen Bonds in Liquid Water Molecular Dynamics Simulation (Tip3P Water Model with CHARMM Force Field), June 25, 2007. self-made with open source visualization software PyMol. http://commons.wikimedia.org/wiki/File:Liquid_water_hydrogen_bond.png.!User:Itub. Diamond and Graphite Samples with Their Respective Structures. The Bottom Right Formation of Carbon Is What Is Known as "Graphene," Characterized by Infinite, Single Atom Sheets of Carbon., March 2007. Self-made derivative work (see below). http://commons.wikimedia.org/wiki/File:Diamond_and_graphite.jpg.
Acknowledgements