163 Hyperglycemia-Induced Hyperplasia of Interstitial Cells of Cajal (ICC) and ICC Stem Cells...
Transcript of 163 Hyperglycemia-Induced Hyperplasia of Interstitial Cells of Cajal (ICC) and ICC Stem Cells...
AG
AA
bst
ract
s
PS: Square pulse(s); E +C: Epinephrine (5 nM) plus corticosterone (1 μM), 10 min; MRS-2500 1 μM, 10 min; L-NAME 200 μM, 15 min; fIJP: fast IJP; sIJP: slow IJP; * was significantbefore and after treatment (p , 0.05, n = 4).
163
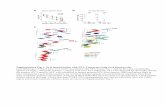
Hyperglycemia-Induced Hyperplasia of Interstitial Cells of Cajal (ICC) andICC Stem Cells (ICC-SC) Is Associated With Accelerated Gastric Emptying inObese Diabetic LeprDb/Db MiceYujiro Hayashi, Yoshitaka Toyomasu, Michael R. Bardsley, Jessica E. Mason, Seth T.Eisenman, Kyoung Moo Choi, David R. Linden, Joseph H. Szurszewski, Brian P. Rubin,Gianrico Farrugia, Tamas Ordog
Background & Aims: ICC depletion is the most common cellular change in diabeticgastroparesis. However, in up to 22% of diabetic patients with gastric symptoms, gastricemptying (GE) is accelerated, rather than delayed. The fate of ICC in these patients isunclear. Previously we reported ICC and ICC-SC hyperplasia in hyperinsulinemic, diabeticLeprdb/db mice. Here, we studied the relationship between GE of solids and ICC/ICC-SCnumbers, and investigated the contribution of insulin/IGF1-dependent Kit ligand (Kitl)expression and hyperglycemia-induced ERK MAPK activation to ICC/ICC-SC hyperplasia.Methods: Leprdb/db (n=32) and age- sex- and strain-matched controls (n=30) were studied13-65 weeks after the onset of diabetes. ICC and ICC-SC were quantified by flow cytometry.Serum insulin was measured by enzyme immunoassay. Oxidative stress was determined bymeasuring serum malondialdehyde (MDA) and gastric 8OHdG. GE of solids was analyzedby 13C breath test. Gene expression was studied by qRT-PCR and WB. Effects of high glucosewere investigated in murine gastric ICC primary cultures, a conditionally immortalized cellline derived from murine gastric ICC (ICL2A), and in an ICC-SC line isolated from themouse stomach (2XSCS2F10). ERKMAPK signaling was evaluated byWB and pharmacologi-cal inhibition of MAPKK with PD98059. Cell proliferation and apoptosis were determinedbyMTS assay andCaspase Glo-3/7 assay, respectively. Results: Leprdb/dbmice had significantlyhigher blood glucose (median[IQR]: 515[462;577] vs. 124[115;134] mg/dL; P,0.001) andserum insulin levels. Serum MDA and gastric 8OHdG were moderately increased (2.4- and2.2-fold, respectively, P,0.001). In Leprdb/db mice, ICC and ICC-SC increased 2.0±0.1-fold(P,0.001) and 1.5±0.3-fold (P,0.05), respectively, and GE was accelerated (T1/2: 67±10vs. 100±11 min; P,0.05). GE was similarly accelerated in KitK641E/+ mice with generalizedICC hyperplasia due to an activating Kit mutation. In Leprdb/db mice, total/soluble Kitl mRNAand Kitl protein were reduced. High glucose (500 mg/dL) significantly increased the numbersof primary ICC and stimulated proliferation and ERK MAPK phosphorylation in ICL2A and2XSCS2F10 cells. PD98059 (20 μM) inhibited cell proliferation induced by high glucosewithout affecting basal proliferation. In contrast, osmotic stress induced by mannitol hadno effect on ICC numbers, cell proliferation and ERK MAPK phosphorylation. ICC andICC-SC were resistant to apoptosis induced by hyperglycemia at levels seen in Leprdb/db
mice. Conclusions: In the absence of major oxidative stress, hyperglycemia induces ICC-SC and ICC hyperplasia via the ERK MAPK pathway even in the presence of reduced Kitlsignaling. ICC hyperplasia, in turn, leads to accelerated GE and may contribute to gastricsymptoms in a subset of diabetic patients. Grant support: NIH DK58185, DK68055.
164
FODMAP Diet Modulates Visceral Nociception by Changing Gut Microbiotaand Intestinal InflammationShi-Yi Zhou, Pornchai Leelasinjaroen, Xiaoyin Wu, Hui Zhou, Yuanxu Lu, Il Song, ChungOwyang
Foods that are high in FODMAPs (fermentable oligo-, di- and mono-saccharides and polys)may exacerbate symptoms of IBS. Thus a diet with a restricted FODMAP content has recentlygained attention for the treatment of IBS. We hypothesize that FODMAP modulates visceralnocieption by altering gut microbiota and intestinal inflammation. To test this hypothesis,Wistar rats were fed low (0%) (LFM) high (10%) FODMAP (HFM), or regular chow (RC,2% FODMAP) for 2 wks. HFM caused intestinal inflammation characterized by 2.8 and 2.5fold increase in mRNA levels for TNFα and IL6 in the ileal mucosa (P ,0.05). This wasaccompanied by 3 fold increase in gut permeability (0.60 vs 0.18 μg/ml, 4000 Da fluorescentdextran-FITC) and a 27% decrease in transepithelial electrical resistance in the ileal mucosa.HFM increased plasma LPS levels from 0.27 to 0.69 EU/ml, P ,0.05). 454 pyrosequencingof IBS RNA gene revealed HFM caused a significant decrease in unclassified clostridiales(18% vs 70%), Peptostreptococaceal (,1% vs 12%) and Lactobacillaceae (,1% vs 12%)accompanied by a significant increase in Erysipelotrichaceae (69% vs 5%) and Lachnospira-ceae (5% vs ,1%; P,0.05 for all comparison). Behavioral pain studies showed that VMRto 20,40 and 60 mmHg colorectal distension increased by 2.0, 3.6 and 2.8 fold following2 wk of HFM (P,0.05, n=5 in ea gp) suggesting development of visceral hypersensitivity.No difference was observed between LFM and RC. In separate studies, rats subjected to 1hr of water avoidance stress (WAS) for 10 d showed evidence of mucosal inflammationcharacterized by increased numbers of neutrophils and mononuclear cells accompanied byincreased gene expression of IL17 (3.5 fold), IL6 (3 fold) and TNF α (7 fold). This wasaccompanied by a 25% decrease transepithelial electrical resistance (TEER) (in vitro) and a45% decrease in gene expression of the junction protein occludin. Two wks of LFM preventedmucosal inflammation and normalized TEER (40.2+2 V/cm2 vs 33+2 V/cm2 in WAS rats,P,0.05). In contrast, 2 wks HFM further increased mucosal inflammation evoked by WASand worsened TEER in the distal ileum (27.1+2 V/cm2, P,0.05). Pain behavioral studies
S-38AGA Abstracts
showed VMR to 20,40 and 60 mmHg CRD increased by 2.3, 2.5 and 2.1 fold in WAS ratscompared to control (P,0.05). Two wks LFM normalized the VMR to CRD in WAS ratswhereas HFM augmented the increased VMR to CRD evoked by stress. In conclusion, HFMcaused dysbiosis in the gut accompanied by mucosal inflammation, impaired permeabilityand visceral hypersensitivity. In contrast, LFM reduced intestinal inflammation and improvedgut barrier functions in WAS rats. These were accompanied normalization of visceral hyper-sensitivity. These actions may explain the beneficial effects of LFM in treating patientswith IBS.
165
Comparison of Gastric Motor Functions and Satiation in Overweight andObese AdultsAndrea Shin, Michael Camilleri, Andres Acosta Cardenas, Barham K. Abu Dayyeh, MariaI. Vazquez-Roque, Jessica O'Neill, Deborah J. Eckert, Duane D. Burton, Alan R.Zinsmeister
Background: Postprandial change in gastric volume (GV) was previously shown to be anindependent predictor of satiation (maximum caloric load ingested). Satiation may bemediated in part by altered gastric functions such as gastric emptying (GE) and fasting andaccommodation GV. Aim: To compare GE, fasting and postprandial GV, maximum toleratedvolume (MTV), satiation and satiety in overweight and obese adults. Methods: Non-bulimicoverweight with BMI 25-29.99kg/m2, obese with BMI 30-34.99kg/m2, and obese with BMI≥35.0kg/m2 adults weighing ,137kg underwent evaluation of gastric motor functions andsatiation. Participants were otherwise healthy with no history of chronic gastrointestinaldisease. On separate days (in random order), we measured GE of solids and liquids of amixed 300kcal meal by scintigraphy, fasting and postprandial (after 300ml Ensure®) GVby single photon emission computed tomography (99mTc-SPECT), total calories ingested atbuffet meal 4h after SPECT (reflecting satiety), and MTV and aggregate symptom scores(satiation) by a validated 100mm VAS scale after a nutrient drink (Ensure® at 30mL/min).The association with BMI group was assessed using ANCOVA adjusted for gender and age(Dunnett-Hsu test for pairwise comparisons), and univariate associations between actualBMI and measured responses by Spearman correlation. Results: Among 43 males and 110females, there were 50 overweight, 55 BMI ,35k/m2, and 48 BMI ≥35kg/m2, with overallmean (±SEM) age of 38.0y (±0.9). Results are summarized in the table. Initial GE of solids[Tlag (p=0.068), GE 1h (p=0.015), and GE 2h (p=0.044)] tended to be associated with BMIgroup, with significant differences between obese BMI ≥35kg/m2 and overweight groups.BMI group was significantly associated with liquid GE T1/2 (p=0.046), but not solid GE T1/2, fasting GV, or aggregate symptom scores. Associations with BMI and postprandial GVand accommodation were of borderline significance. Overall MTV was associated with BMIgroup; the BMI ≥35kg/m2 group had a lower mean MTV than the BMI 30-34.99kg/m2 andoverweight groups. Increased solid GE at 1h was associated with BMI (rs=0.18, p=0.026).Solid GE Tlag, accommodation, and caloric intake were inversely associated with BMI.Conclusions: BMI ≥35kg/m2 is associated with accelerated early GE of solids and decreasedMTV. BMI is inversely related to accommodation and caloric intake, but the contributionof BMI to the variance is modest. The reduced MTV suggests that BMI ≥35kg/m2 may bepartly attributed to accelerated initial GE solids. A learned increased responsiveness tosatiation signals with cessation of intake may represent a behavioral change in those withBMI ≥35kg/m2. Funding: NIH DK67071
!Overall comparisons by ANCOVA adjusted for age and gender. Two-group comparison byDunnett-Hsu test vs. overweight: @p=0.010; #p=0.042; Δp=0.031; ∞p=0.064; ∑p=0.081.Two-group comparison vs. BMI 30-34.99kg/m2: +p,0.05; Spearman correlation coefficient =rs: *p=0.030; #p=0.016; ^p=0.059; $p=0.073










![Hyperglycemia-induced oxidative stress and heart disease ......and heart failure (HF) in a diabetic state [3, 4]. Chronic hyperglycemia alters the myocardial substrate preference in](https://static.fdocument.org/doc/165x107/60ebf67ef3b32f2f70556515/hyperglycemia-induced-oxidative-stress-and-heart-disease-and-heart-failure.jpg)







