A CONVENIENT PREPARATION OF α,α-DIPHENYLACETOPHENONE
Transcript of A CONVENIENT PREPARATION OF α,α-DIPHENYLACETOPHENONE

This article was downloaded by: [University of North Carolina Charlotte]On: 07 August 2013, At: 10:25Publisher: Taylor & FrancisInforma Ltd Registered in England and Wales Registered Number: 1072954Registered office: Mortimer House, 37-41 Mortimer Street, London W1T 3JH, UK
Organic Preparations andProcedures International: The NewJournal for Organic SynthesisPublication details, including instructions for authors andsubscription information:http://www.tandfonline.com/loi/uopp20
A CONVENIENT PREPARATION OFα,α-DIPHENYLACETOPHENONEN. Sbarbati Nudelman a & A. A. Vitale aa Dept. de Quimica Organica, Facultad de Ciencias Exactas yNaturales, Universidad de Buenos Aires, 1428, Buenos Aires,ARGENTINAPublished online: 12 Feb 2009.
To cite this article: N. Sbarbati Nudelman & A. A. Vitale (1981) A CONVENIENT PREPARATIONOF α,α-DIPHENYLACETOPHENONE, Organic Preparations and Procedures International: TheNew Journal for Organic Synthesis, 13:2, 144-149
To link to this article: http://dx.doi.org/10.1080/00304948109356114
PLEASE SCROLL DOWN FOR ARTICLE
Taylor & Francis makes every effort to ensure the accuracy of all the information(the “Content”) contained in the publications on our platform. However, Taylor& Francis, our agents, and our licensors make no representations or warrantieswhatsoever as to the accuracy, completeness, or suitability for any purpose of theContent. Any opinions and views expressed in this publication are the opinions andviews of the authors, and are not the views of or endorsed by Taylor & Francis. Theaccuracy of the Content should not be relied upon and should be independentlyverified with primary sources of information. Taylor and Francis shall not be liablefor any losses, actions, claims, proceedings, demands, costs, expenses, damages,and other liabilities whatsoever or howsoever caused arising directly or indirectly inconnection with, in relation to or arising out of the use of the Content.
This article may be used for research, teaching, and private study purposes. Anysubstantial or systematic reproduction, redistribution, reselling, loan, sub-licensing,systematic supply, or distribution in any form to anyone is expressly forbidden.

Terms & Conditions of access and use can be found at http://www.tandfonline.com/page/terms-and-conditions
Dow
nloa
ded
by [
Uni
vers
ity o
f N
orth
Car
olin
a C
harl
otte
] at
10:
25 0
7 A
ugus
t 201
3

OPPI BRIEFS
!s, 3, C O C H j ) . -
6 . The cis a x i a l - e q u a t o r i a l r e l a t i o n s h i p f o r t h e hydrogens
a t t h e 1 and 6 p o s i t i o n s o f p u r i f i e d 1 w a s conf i rmed by
t h e magnitude of t h e i r c o u p l i n g c o n s t a n t (J = 4.03 Hz) a s
a s c e r t a i n e d by doub le resonance expe r imen t s . Composed of
1 3 .sharp l i n e s , t h e 13C NMR of - 1 is also i n harmony w i t h
t h e p r e s e n c e of a s i n g l e s t e r e o i s o m e r . W e a r e assuming
t h a t t h e bulky 3-buten-2-one s i d e c h a i n of 1 o c c u p i e s an
e q u a t o r i a l r i n g p o s i t i o n , t h e r e b y p r e c l u d i n g t h e p o s s i -
b i l i t y of a t r a n s e q u a t o r i a l - e q u a t o r i a l r e l a t i o n s h i p f o r
t h e 1 and 6 r i n g p o s i t i o n p r o t o n s o f A. 7. T h i s y i e l d of p u r e 1 r e p r e s e n t s a 57% recove ry o f t h e a-
v a i l a b l e 1 i n t h e mix tu re . W e have found t h a t a less con-
s e r v a t i v e c o l l e c t i o n of 1. by HPLC s e r i o u s l y compromises
i t s p u r i t y .
A CONVENIENT PREPARATION OF a.a-DIPHENYLACETOPHENONE
Submit ted b N . Sbarbati Nudelman" and A. A. V i t a l e 7 - i 7 i m r Y
Dept. de Quimica Organica F a c u l t a d de C i e n c i a s E x a c t a s y N a t u r a l e s Un ive r s idad de Buenos A i r e s 1428 Buenos A i r e s . ARGENTINA
Although s e v e r a l m e c h a n i s t i c s t u d i e s and s y n t h e t i c a p p l i -
c a t i o n s o f a l k a l i a r o m a t i c k e t y l ~ l - ~ have been r e c e n t l y pub-
l i s h e d , t h e r e have been n 3 f u r t h e r s t u d y of t h e r e a c t i o n o f
p h e n y l l i t h i u m w i t h ca rbon monoxide s i n c e t h e work of White-
144
Dow
nloa
ded
by [
Uni
vers
ity o
f N
orth
Car
olin
a C
harl
otte
] at
10:
25 0
7 A
ugus
t 201
3

OPPI BRIEFS
s i d e s , e t al.’
to be an i n t e r m e d i a t e b u t s e v e r a l mechan i s t i c q u e s t i o n s re-
mained unreso lved .
I n t h a t paper t h e benzophenone k e t y l w a s shown
As p a r t o f a d e t a i l e d i n v e s t i g a t i o n o f t h e mechanism of
t h e r e a c t i o n , w e have e s t a b l i s h e d c o n d i t i o n s which accoun t f o r
t h e t o t a l f a t e o f p h e n y l l i t h i u m and, s econd ly , minimize t h e
fo rma t ion o f by-products . Only f o u r p roduc t s were formed:
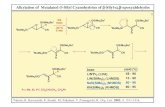
a,a-diphenylacetophenone ( I ) i s t h e major p roduc t , benzophe-
none (11) , benzoin (111) and a, a-diphenyl-a-hydroxyacetophenone
( I V ) . 0 OOH OOH
II I 0 II U II I
PhLi PhCCHPh2 + PhCPh + PhCCHPh + PhCCPh2
I I1 I11 I V
S o l v e n t and t empera tu re e f f e c t s , among o t h e r v a r i a b l e s ,
were s t u d i e d . The s o l v e n t s t e s t e d w e r e : d i e t h y l e t h e r , ~f-
hexane, t e t r a h y d r o f u r a n e and dimethoxyethane , b u t t h e b e s t
y i e l d s were o b t a i n e d u s i n g p h e n y l l i t h i u m as a f i n e powder,
w i t h no s o l v e n t . Some r e p r e s e n t a t i v e r e s u l t s are g a t h e r e d i n
t h e Table.
TABLE.- P roduc t Y i e l d ( % ) from S o l i d PhLi and COa
Temp. (OC) PhLi (mole) I I1 I11 I V
0 4 . 4 4 2 33 15 9 0 5.0 44 33 1 3 9
25 6 . 1 56 25 11 7 25 3 . 7 6 3 20 1 0 6 .5 60 4 . 2 70 19 7 3 60 4.9 78 14 5 2 - l lOb 5.0 94 1 .5 -
a) The y i e l d s r e p r e s e n t % conver s ion . b) 4.5% o f p h e n y l l i t h i - um w a s c o n v e r t e d t o t r i p h e n y l c a r b i n o l .
1 4 5
Dow
nloa
ded
by [
Uni
vers
ity o
f N
orth
Car
olin
a C
harl
otte
] at
10:
25 0
7 A
ugus
t 201
3

OPPI BRIEFS
A s it c a n be obse rved t h e conve r s ion to I i n c r e a s e s w i t h
t h e t empera tu re o f r e a c t i o n w h i l e t h e y i e l d o f 11 d e c r e a s e s ,
a s it h a s been observed i n d i e t h y l e t h e r s ~ l u t i o n . ~ The re-
a c t i o n s l e a d i n g to p r o d u c t s X I 1 and I V w e r e a l s o minimized a t
h i g h t empera tu res . All t h e r e p o r t e d y i e l d s w e r e o b t a i n e d by
g l p c ; f o r t h e r e a c t i o n s c a r r i e d o u t a t 60° and l l O o , I was a l -
so i s o l a t e d by s t a n d a r d t e c h n i q u e s .
I t i s wor th ment ioning t h a t t h e p r e v i o u s p r e p a r a t i o n s o f
I from benzoin6 ( y i e l d : 51%, 4 1 % o v e r - a l l s t a r t i n g from ben-
za ldehyde ) or from t r i p h e n y l e t h y l e n e 8 ( y i e l d : 59%, 35% ove r -
a l l from b e n z y l c h l o r i d e ) , are more l a b o r i o u s t h a n t h a t des -
c r i b e d h e r e . Fur thermore , t h e s y n t h e t i c c a p a b i l i t i e s o f t h e
a l k a l i a r o m a t i c k e t y l s are v e r y w e l l known;" t h e r e a c t i o n i s
now b e i n g examined i n an a t t e m p t t o i n t r o d u c e o r g a n i c g roups
b e f o r e t h e h y d r o l y t i c quenching. P r e l i m i n a r y r u n s w i t h b u t y l
are p romis ing b u t methyl i o d i d e t r e a t m e n t gave a m i x t u r e o f
p r o d u c t s as p r e v i o u s l y obse rved w i t h s imi l a r a n i o n s .
7a
9
2
EXPERIMENTAL
Mel t ing p o i n t s are u n c o r r e c t e d . A l l compounds r e p o r t e d h e r e w e r e f u l l y c h a r a c t e r i z e d by m a s s spec t romet ry ( u s i n g a Var i an M a t CH7 s p e c t r o m e t e r ) , i n f r a r e d (de t e rmined on a P e r - k i n E l m e r 137 spec t ropho tomete r ) , u l t r a v i o l e t ( r e c o r d e d on a Beckman DK2A spec t ropho tomete r ) and n u c l e a r magnet ic reson- ance (de t e rmined on a Var i an A-60 s p e c t r o m e t e r ) s p e c t r o s c o p y and showed s p e c t r a l c h a r a c t e r i s t i c s c o n s i s t e n t w i t h t h e spec - t r a o f a u t h e n t i c samples . The g l p c a n a l y s e s w e r e c a r r i e d o u t on a 5830 Hewlet t -Packard G a s Chromatograph. Reac t ions invo lv - i n g p h e n y l l i t h i u m w e r e c a r r i e d o u t u s i n g s t a n d a r d t e c h n i q u e s f o r m a n i p u l a t i o n of oxygen and w a t e r - s e n s i t i v e compounds.
P h e n y l l i t h i u m was p r e p a r e d by a method adap ted from t h a t of S c h l o s s e r and Ladenberger ;12 no d i f f e r e n c e was obse rved by u s i n g f r e s h l y p r e p a r e d b u t y l l i t h i u m o r t h e commercial s o l u - t i o n i n E- exane. Carbon monoxide was g e n e r a t e d by s t a n d a r d proceduresSb and p u r i f i e d by p a s s i n g through a column o f po- t a s s i u m hydroxide and t h e n bubb l ing through a s o l u t i o n of ben- zophenone k e t y l i n t o l u e n e .
14 6
Dow
nloa
ded
by [
Uni
vers
ity o
f N
orth
Car
olin
a C
harl
otte
] at
10:
25 0
7 A
ugus
t 201
3

OPPI BRIEFS
Reaction of Phenyl l i thium with Carbon Monoxide.- The phenyl l i -
thium contained i n a tube capped with a No-air s topper w a s ex-
posed to carbon monoxide (1 atm p r e s s u r e ) . The white s o l i d
turned immediately t o pink, then to red and when t h e gas ab-
s o r p t i o n had ceased was a brownish purp le powder. n-Undecane
was added as i n t e r n a l g l p c s tandard , then g-hexane to form a h o -
mogeneous suspension (10 m l ) . A 1 . 0 m l a l i q u o t of t h e r e a c t i o n
mixture w a s quenched with w a t e r , e x t r a c t e d wi th e t h e r and t h e
d r i e d (MgS04) e t h e r l a y e r was analyzed by glpc.
I s o l a t i o n and Charac te r iza t ion of t h e Products.- The remaining
s o l u t i o n was worked up i n a s i m i l a r way and t h e e t h e r l a y e r
w a s d i s t i l l e d a t reduced pressure . The white r e s i d u e w a s
t r e a t e d with methanol and t h e c r y s t a l s c o l l e c t e d . A y i e l d of
60% of I , mp. 137' was obta ined from t h e r e a c t i o n mixture .car-
r i e d o u t a t 60°. The methanolic s o l u t i o n w a s passed through
a column of s i l i c a g e l with benzene-methanol mixture a s e l u e n t .
F r a c t i o n s conta in ing I1 and I11 were collected and charac te r -
i z e d by t h e i r mp., s p e c t r a and t l c behaviour. Comp. I V i s
produced only i n small amount and w a s i d e n t i f i e d by glpc-ms.
Product Balance by g1pc.- The r e a c t i o n mixture w a s analyzed
by g lpc on a 3% SE 30 on Chromosorb W column a t 50-250°.
t h i s column benzoin and b e n z i l have t h e same r e t e n t i o n t i m e
b u t only benzoin was i s o l a t e d by column chromatography and
f u l l y charac te r ized .
Standard Prepara t ion of I.- n-Butyll i thium ( 1 0 m l of a 1 . 2 N
hexane s o l u t i o n ) was t r a n s f e r r e d by a s y r i n g e to a 20 m l cen-
t r i f u g e tube ( n i t r o g e n atm) conta in ing s e v e r a l g l a s s p e a r l s ,
and capped wi th a No-air s topper . Iodobenzene (1.6 m l ) w a s
On
147
Dow
nloa
ded
by [
Uni
vers
ity o
f N
orth
Car
olin
a C
harl
otte
] at
10:
25 0
7 A
ugus
t 201
3

OPPI BRIEFS
added dropwise a n d PhLi p r e c i p i t a t e d as w h i t e c r y s t a l s , which
w e r e t h r i c e washed w i t h fi-hexane added by a s y r i n g e a n d dr ied
a t 60° and 1 0 mm Hg p r e s s u r e .
c a r b o n monoxide (1 a t m p r e s s u r e ) w a s p a s s e d t h r o u g h a l e n g t h o f
t y g o n t u b i n g and hypodermic n e e d l e . F a s t g a s a b s o r p t i o n o c u r r e d
i n t h e f i r s t 1 0 m i n t t h e n i t s lowed and c e a s e d c o m p l e t e l y ( 8
mmole of CO) i n 4 h r . The r e s u l t i n g r e a c t i o n m i x t u r e w a s a l -
lowed t o cool t o room t e m p e r a t u r e , n e x t i t w a s s u s p e n d e d i n ;-
hexane (10 m l ) , h y d r o l y z e d w i t h w a t e r ( 5 m l ) , e x t r a c t e d w i t h
d i e t h y l e t h e r a n d d r i ed over magnesium s u l f a t e . D i s t i l l a t i o n
o f t h e e t h e r a t 30° and r e d u c e d p r e s s u r e r e n d e r e d 1 . 0 3 g ( 9 4 %
y i e l d ) o f I , c r y s t a l l i z a t i o n f rom m e t h a n o l gave mp. 137O.
Acknowledgements.- The a u t h o r s are i n d e b t e d t o t h e C o n s e j o
N a c i o n a l d e I n v e s t i g a c i o n e s C i e n t i f i c a s y T e c n i c a s ( A r g e n t i n a ) ,
t o t h e SECYT ( A r g e n t i n a ) a n d t o t h e O r g a n i z a t i o n of American
S t a t e s f o r f i n a n c i a l s u p p o r t . UMYMFOR ( A r g e n t i n a ) is acknow-
l e d g e d f o r t h e g l p c a n d m a s s s p e c t r a .
The t u b e w a s h e a t e d a t llOo and
1.
2 .
3.
4 .
5.
6.
7.
8 .
REFERENCES
J. G. S m i t h a n d D . J. M i t c h e l l , J. Am. Chem. SOC., 99, 5045 ( 1 9 7 7 ) .
J . F. Garst a n d C . D. S m i t h , i b i d . , 98, 1520 ( 1 9 7 6 ) .
H . W. Wang, G. L e v i n a n d M. Szwarc , i b i d . , 99, 5056 ( 1 9 7 7 ) .
S. M. R o s e n f e l d , T e t r a h e d r o n L e t t . , 2655 ( 1 9 7 8 ) .
L. S. Trzupek, T. L. N e w i r t h , E . G . K e l l y , N . S. Nudelman
a n d G. M. W h i t e s i d e s , J. Am. Chem. SOC., 95, 8118 ( 1 9 7 3 ) .
0. P o l a n s k y , E . S c h i n z e l and F. Wesse ly , Monatsh Chem., 87, 2 4 ( 1 9 5 6 ) .
A. Vogel , " P r a c t i c a l O r g a n i c C h e m i s t r y " , 3 r d . ed. , Longman,
London, a ) p. 714; b) p . 185 ( 1 9 5 6 ) .
A. Cope, P. A. Trumbul l and R. E . T r u m b u l l , J. Am. Chem.
SOC., 80, 2844 ( 1 9 5 8 ) .
148
Dow
nloa
ded
by [
Uni
vers
ity o
f N
orth
Car
olin
a C
harl
otte
] at
10:
25 0
7 A
ugus
t 201
3

OPPI BRIEFS
9. H . Adkins and W. Zartman, Org. Syn., 17, 89 (1937). 10. S. Selman and J. F. Eastham, J. Org. Chem., 3c), 3804
(1956): 0. Halpern and C. njerassi , J. Am. Chem. SOC. , - 81, 493 (1959).
11. D. F. S h r i v e r , "The Manipulation of Air-Sensi t ive Com-
pounds", M c - G r a w H i l l , N e w York, N. Y . , 1969, C h . 7. 12. M. Sch losse r and V. Ladenberger, J. Orqanomet. Chem., 2,
193 (1967).
AN IMPROVED PREPARATION O F CORONENE
Submitted b A. G. HolDa and E. J. Eisenbraun* d Department of Chemistry Oklahoma S t a t e Un ive r s i ty S t i l l w a t e r , OK 74078
Syn thes i s of coronene i s g e n e r a l l y on a mg scale. Our
a t t e m p t s t o i n c r e a s e t h e scale w e r e f r u s t a t e d u n t i l w e developed
a h e a t e r and improved glassware' which enabled s u c c e s s f u l l y
c a r r y i n g o u t t h e , r e a c t i o n a t t h e r e q u i r e d temperature. Pre-
0 2 v ious ly r epor t ed y i e l d s of coronene were 66% (mp. 425-428 )
and 73% crude with 18% p u r i f i e d . We c o n s i s t e n t l y ob ta ined
pure coronene i n 80% y i e l d , mp. 434-438O (uncorr . ) on a gram
scale.
I1 EXPERIMENTAL I
Melting p o i n t s w e r e determined on Mel-temp c a p i l l a r y m e l t - i n g p o i n t appa ra tus w i t h a 90-510° thermometer from H-B i n s t r u -
149
Dow
nloa
ded
by [
Uni
vers
ity o
f N
orth
Car
olin
a C
harl
otte
] at
10:
25 0
7 A
ugus
t 201
3