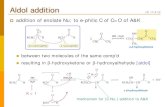
Seminário Importância Da Condensação de Claisen e Condensação Aldólica
Rainer Ludwig Claisen (1851-1930) Developed the Claisen Condensation in 1887.
-
Upload
kaitlyn-gallagher -
Category
Documents
-
view
244 -
download
0
Transcript of Rainer Ludwig Claisen (1851-1930) Developed the Claisen Condensation in 1887.

Rainer Ludwig Claisen (1851-1930)
Developed the “Claisen Condensation” in 1887


What are the building blocks?

sp2 orbitals
p orbitalsπ bond
sp3 orbitals
α hydrogens

αα hydrogens hydrogens
Sites for Sites for nucleophilic nucleophilic attacksattacks
High Electronegativity of Oxygen

Resonance Energy“Stabilization energy”

Ethyl propanoate
Formation of the enolate
Condensationwith ethyl
benzoate to form β-keto ester
Et -O - C - CH2 – CH3
O||
Loss of 2nd alpha hydrogen on β-keto ester
Et -O - C - CH – CH3
O|| -

Ethyl Propanoate
0.040.04
pKa of alpha hydrogens ≈ 24 or Ka ≈ 10-24

Bond Angles
120o
sp2 orbitals109.5o
sp3 orbitals
180o
sp orbitals
90o
p orbitals

Overview of the Claisen Condensation Reaction
Ethyl Propanoate
Ethoxide ionEthyl Benzoate
1. Ethoxide ion acting as a strong base deprotonates ethyl propanoate.
2. The enolate of ethyl propanoate is formed.
3. The enolate of ethyl propanoate is a strong nucleophile that attacks the partially positive carbon of the carbonyl group on ethyl benzoate.
4. The alpha substitution makes this a carbonyl condensation reaction.
5. A β-keto ester is formed.
+
6. The driving force for this reaction is the transfer of the second alpha hydrogen to the ethoxide ion. This is exothermic.

Deprotonation of Ethyl Propanoate
ResonanceThe enolate ion is a strong nucleophile and will attack the partially positive carbon of a carbonyl group.
+
Ethyl PropanoateEthyl Propanoate
Ethyl Benzoate
Enolate ofEnolate of

Self condensation of ethyl propanoate



Rainer Ludwig Claisen (1851-1930)
Developed the “Claisen Condensation” in 1887