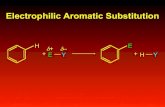
Effect of Selenium Substitution on Intersystem Crossing in π ...
Mechanism Summary for AS AQA Chemistry - … Summary for AS AQA Chemistry Nucleophilic Substitution...
-
Upload
truongduong -
Category
Documents
-
view
217 -
download
1
Transcript of Mechanism Summary for AS AQA Chemistry - … Summary for AS AQA Chemistry Nucleophilic Substitution...
CH3 C
H
H
Br CH3 C
H
H
OH-HO:
+ :Br -δ+ δ-
CH3 C
H
H
CNCH3 C
H
H
Br-NC:
+ :Br -δ+ δ-
CH2 C
H
H
BrCH3
CH2 C
H
H
NH2CH3
CH2 C
H
H
N+
CH3
H
H
H
3HN:
δ+ δ-
+ NH4Br
:OH-
C C
H
H
CH3
Br
H
H C C
H
CH3
H
H
+ Br - + H2O:NH3
:Br -
C C
H
H
H
H
C C
H
H
Br Br
H
H
C+
C
Br
H
HH
H
Br
Brδ+
δ-
:Br -
C+
C
H
CH3
HH
CH3
C C
H
CH3
HH
CH3
Br
C C CH3
HH
CH3
δ+
δ- :Br -
H
Br
H
OSO2OH
C C H
HH
CH3
δ+
δ-
-:OSO2OH
C+
C H
HH
CH3
H
C C H
HH
CH3
HOSO2OH
STEP ONE Initiation
Br2 2Br .Essential condition: UV light
CH3CH3 + Br. HBr + CH3CH2.
STEP TWO Propagation
CH3CH2. + Br2 CH3CH2Br + Br.
STEP THREE TerminationCH3CH2
. + Br. CH3CH2Br
CH3CH2. + CH3CH2
. CH3CH2CH2CH3
Mechanism Summary for AS AQA Chemistry
Nucleophilic Substitution of Halogenoalkaneswith aqueous hydroxide ions.
Nucleophilic Substitution of Halogenoalkaneswith cyanide ions.
Nucleophilic Substitution of Halogenoalkanes with ammoniaElimination of Halogenoalkanes with ethanolichydroxide ions
Electrophilic Addition of Alkenes with Bromine Electrophilic Addition of Alkenes with hydrogenbromide
Electrophilic Addition of Alkenes with sulphuric acid
Free Radical Substitution of Alkanes with Bromine
:CH3 C CH3
O H
H
CH3 C CH3
O+
H
H
H
C C+
CH3
H
H
H
H
H+H+
C C CH3
H
H
H
Acid catalysed elimination mechanism: alcoholsalkenes
The H+ comes from the conc H2SO4 or conc H3PO4
C C
HH
H H
C C+
H
H
H
H
H
OH
H
C C
O+
H
H
H
H
H
H
H
C C
O
H
H
H
H
H
HH+
H+
Acid catalysed addition mechanism for hydration of ethene
The H+ comes from the conc H3PO4
Alcoholic NH3heat under pressurenucleophilic substitution
1 H2SO42 H2O warmElectrophilicAddition
KOH aqueousheat under reflux
nucleophilic substitution
Br2, Cl2 UV lightFree radicalSubstitution
KOH aqueousheat under refluxnucleophilic substitution
amine
alkane
halogenoalkane
alkene
alcohol
ketonealdehyde
carboxylic acid
diol
dihalogenoalkane
conc H2SO4 orconc H3PO4Elimination
If primary alcohol or aldehydeK2Cr2O7/H+
heat under reflux+ excess oxidising agent
Oxidation
nitrile
KCN in alcoholheat under refluxnucleophilic substitution
poly(alkene)
high pressurecatalyst
AS Reactions- Summary