Lesson 1 X-Rays & Diffraction -...
Transcript of Lesson 1 X-Rays & Diffraction -...

Lesson 1
X-Rays & Diffraction
Nicola Döbelin
RMS Foundation, Bettlach, Switzerland
June 07 – 09, 2017, Oslo, N

Electromagnetic Spectrum
2
X rays:
Wavelength λ: 0.01 – 10 nm
Energy: 100 eV – 100 keV
Generation of X-radiation:
Shoot electrons on matter
Interatomic distances in crystals:
typically 0.15 – 0.4 nm
Interference phenomena only
for features ≈ λ

X-ray Tube
3
e‒
Filament (W)
Target (Cu, Mo, Fe, Co, ...)
Be window
Acceleration
Voltage
Va
cu
um
Filament
Current

X-ray Tube
4
e‒
Filament (W)
Target (Cu, Mo, Fe, Co, ...)
Point focus
Line focus

Generation of X-rays
5
Accelerated electron impinges on matter:
Electron is deflected and decelerated by the atomic nucleus.
(Inelastic scattering)
Deflected electron emits electromagnetic radiation.
Wavelength depends on the loss of energy.
Bremsstrahlung (Deceleration radiation)

Bremsstrahlung
6
Wavelength (nm)
Inte
nsity
0.00 0.05 0.10 0.15 0.20
20 kV, 20 mA
30 kV, 20 mA
40 kV, 20 mA
Continuous spectrum
0.25 0.30
Acceleration Voltage:
Speed / Energy of Electrons

Bremsstrahlung
7
Wavelength (nm)
Inte
nsity
0.00 0.05 0.10 0.15 0.20
30 kV, 20 mA
Continuous spectrum
0.25 0.30
30 kV, 30 mA
30 kV, 40 mA Generator Current:
Number of Electrons

Characteristic Radiation
8
K L M
Eb (eV)
0
76
122
933 952
1097
8979
M4,5 (3d)
M2,3 (3p)
M1 (3s)
L3 (2p3/2)
L1 (2p1/2)
L1 (2s)
K1 (1s)
Kα1 Kα2 Kβ
Cu
Wavelength of Kα1, Kα2, Kβ, Lα... are characteristic for the atomic species.

X-rays: Spectrum
9
Wavelength (nm)
Inte
nsity
0.00 0.05 0.10 0.15 0.20 0.25 0.30
Cu
Kα1
Kα2
Kβ
Kα1
Kα2
Kβ
Mo

Old X-ray tubes
10
Caution:
Beryllium is toxic & carcinogenic!
- Never touch the windows!
- Use appropriate covers!
Lifetime of a few years:
- Vacuum decreases
loss of intensity
- Tungsten from filament deposits
on target
contaminated spectrum (characteristic
W spectrum starts to appear)
- Monitor the intensity
- Replace old tubes

X-rays: Summary
11
• Generated in an X-ray tube
• Spectrum contains Bremsstrahlung (continuous) and
characteristic radiation (Kα1, Kα2, Kβ) of target material
• Tube is characterized by: • Target material
• Size and shape of target
• Aceleration voltage and
current

Diffraction Basics
12
Interaction of X-rays with matter:
- Absorption (photoelectric effect, giving rise to fluorescence)
- Elastic scattering (Thomson scattering)
- Inelastic scattering (Compton scattering)
Absorption Photoelectric effect, Fluorescence
1. Absorption and ionization
2. Relaxation and emission of characteristic radiation
CuKα1
FeKα1
Fe atom

Elastic Scattering
13
Electron oscillates in the electric field,
emits photons of the same wavelength as
the incoming radiation (λs = λp).
λp
λs
CuKα1
CuKα1
Fe atom
Secondary wave is in phase
(+ 180°) with primary wave.

Crystal Lattice
14
Crystal: Periodic arrangement of atoms/ions/molecules in 3 dimensions.
Electrons of each atom become a source of
scattered radiation (spherical waves)

15
Interference pattern
n=2
+ =
Positive / constructive
interference
Amplification
+ =
Negative / destructive
interference
Extinction

16
Interference pattern
n=4 n=8 n=12 n=25 n=50

Bragg’s Law
17
n ∙ λ = 2 ∙ d ∙ sin(θ)
d
λ
θ θ δ
2δ = n ∙ λ δ = d ∙ sin(θ)
d = (n ∙ λ) / (2 ∙ sin(θ))

Lattice Planes and Miller Indices
18
a
b
Definition:
A lattice plane is a plane
which intersects atoms of
a unit cell across the whole
3‐dimensional lattice.
d(100)
d(010)
d(110)
d(-210)
- Each lattice plane
generates a diffraction
peak.
- The 2θ angle of the
peak depends on the
plane’s d-spacing.
- Diffraction peaks can
be labelled with the
plane’s Miller index.

Bragg’s Law
19
CuKα1 = 0.154056 nm
a = 0.2 nm
b = 0.5 nm
b a
b
a
2θ = 45.30°
2θ = 17.72°
d = 0.2 nm
d = 0.5 nm
θ = 22.65°
θ = 8.86°

Single Crystal
20
A single crystal must be rotated
to bring each lattice plane in
diffraction condition.

Polycrystals, Powders
21
In an ideal powder every
possible orientation of
crystals occurs.
In a random powder
no orientation is preferred.
In an ideal powder all possible diffraction peaks
are generated, regardless of sample orientation.

Diffraction Cones
22
Diffraction at an angle 2θ° from
the primary beam
All possible rays form a cone = diffraction cone = Debye cone

Diffraction Cones
23
(120)
(100)
(010)
One Debye Cone for each lattice plane spacing (d value)
Powder sample:

Debye Ring
24
Gra
y V
alu
e
2 Angle

Powder Diffractometer
25
X-ray tube
Primary Beam
Powder
Sample
Diffraction Cones
«Secondary Beams»
X-ray Detector scanning X-ray intensity
vs. 2θ angle

Powder Diffraction Pattern
26
10 20 30 40 50 60
0
500
1000
1500
2000
Inte
nsity (
cts
)
Diffraction Angle (°2)
Lesson 2: All about powder diffractometers

Monochromatic X-radiation
27
n ∙ λ = 2 ∙ d ∙ sin(θ)
http://fineartamerica.com
Diffraction angle θ depends on wavelength λ:
Polychromatic X-ray Beam
We need monochromatic X-radiation!

28
Monochromatic X-Radiation
Cu Radiation
Kβ absorption filter
filtered Radiation
Ideally: Isolate Kα1
Reality: Suppress Kβ and Bremsstrahlung
Wavelength (nm)
Inte
nsity
0.00 0.05 0.10 0.15 0.20 0.25 0.30
Cu
Kα1
Kα2
Kβ
Bremsstrahlung Cu Radiation
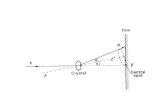
Monochromator Crystal
(Graphite, d = 0.3352 nm)
CuKα1/2 Radiation
n ∙ λ = 2 ∙ d ∙ sin(θ)
Cu Radiation Energy dispersive
Detector
Digital filtering

Kβ-filtered Diffraction Pattern
29
27 28 29 30 31 32
0
100000
200000
300000
400000
500000In
ten
sity
Diffraction Angle (°2)
CuKα1
CuKα2
LaB6 pattern, Cu radiation, Ni filter in primary beam (Bruker D8 Advance)

Kβ-filtered Diffraction Pattern
30
27 28 29 30 31 32
0
2000
4000
6000
8000
10000
12000
14000
16000
18000
20000
Inte
nsity
Diffraction Angle (°2)
CuKβ
CuKα1 & CuKα2 duplet
Remaining Bremsstrahlung
Absorption Edge
Impurity
CuKα Satellites
(= CuKα3)
LaB6 pattern, Cu radiation, Ni filter in primary beam (Bruker D8 Advance)

Graphite Monochromator
31
27 28 29 30 31 32
0
1000
2000
3000
4000
5000
6000
7000
8000
9000
10000In
ten
sity
Diffraction Angle (°2)
CuKα1
CuKα2
LaB6 pattern, Cu radiation, Graphite monochromator, secondary beam (PANalytical CubiX3)

Graphite Monochromator
32
27 28 29 30 31 32
0
100
200
300
400
500In
ten
sity
Diffraction Angle (°2)
CuKα1 & CuKα2 duplet
CuKα Satellites
(= CuKα3)
LaB6 pattern, Cu radiation, Graphite monochromator, secondary beam (PANalytical CubiX3)

31 32 33 34 35 36
0
10000
20000
30000
40000
50000
taken from file NIST-SRM1976b-150105-03.brml
Inte
nsity (
co
un
ts)
Diffraction Angle (°2)
Energy-Dispersive Detector
33
Al2O3 pattern, Cu radiation, LynxEyeXE detector (Bruker D8 Advance)
CuKα1
CuKα2

31 32 33 34 35 36
0
1000
2000
3000
4000
5000
taken from file NIST-SRM1976b-150105-03.brml
Inte
nsity (
co
un
ts)
Diffraction Angle (°2)
Energy-Dispersive Detector
34
CuKβ
CuKα1 & CuKα2 duplet
Edge of
Energy Window
Al2O3 pattern, Cu radiation, LynxEyeXE detector (Bruker D8 Advance)

31 32 33 34 35 36
0
5000
10000
15000
20000
25000
30000
taken from file NIST-SRM1976b-150106-01-NiFilter.brml
Inte
nsity (
co
un
ts)
Diffraction Angle (°2)
Energy-Dispersive Detector + Kβ filter
35
CuKα1
CuKα2
50% loss of intensity
(0.0125 mm Ni filter)
Al2O3 pattern, Cu radiation, LynxEyeXE detector (Bruker D8 Advance)

31 32 33 34 35 36
0
500
1000
1500
2000
2500
3000
3500
4000
4500
5000
taken from file NIST-SRM1976b-150106-01-NiFilter.brml
Inte
nsity (
co
un
ts)
Diffraction Angle (°2)
Energy-Dispersive Detector + Kβ filter
36
CuKα1 & CuKα2 duplet
Al2O3 pattern, Cu radiation, LynxEyeXE detector (Bruker D8 Advance)
Edge of
Energy Window
50% loss of intensity
(0.0125 mm Ni filter)

Monochromators
37
Kβ Filter:
Selectively suppresses Kβ
and parts of Bremsstrahlung
Monochromator crystal and
energy dispersive detector:
Suppress everything BUT Kα
Important difference for
fluorescent samples
«Notch filter»
«Bandpass filter»

38
Ni-Filtered Cu Radiation, Fe Fluorescence
10 20 30 40 50 60 70 80
0
500
1000
1500
2000
G:\Auftr_Proj\S-Auftraege\2012\S12_0008 XRD Course Riga\Paper Andris\Paper2\XRD-Fluorescence.opj
Inte
nsity [cts
]
Diffraction Angle [°2]
T0
T05
T10
T25
T50
T75
CuK radiation, Ni-filternot stacked
10 20 30 40 50 60 70 80
0
500
1000
1500
2000
G:\Auftr_Proj\S-Auftraege\2012\S12_0008 XRD Course Riga\Paper Andris\Paper2\XRD-Fluorescence.opj
Inte
nsity [cts
]
Diffraction Angle [°2]
T0
T05
T10
T25
T50
T75
CuK radiation, Ni-filternot stacked
10 20 30 40 50 60 70 80
0
500
1000
1500
2000
G:\Auftr_Proj\S-Auftraege\2012\S12_0008 XRD Course Riga\Paper Andris\Paper2\XRD-Fluorescence.opj
Inte
nsity [cts
]
Diffraction Angle [°2]
T0
T05
T10
T25
T50
T75
CuK radiation, Ni-filternot stacked
10 20 30 40 50 60 70 80
0
500
1000
1500
2000
G:\Auftr_Proj\S-Auftraege\2012\S12_0008 XRD Course Riga\Paper Andris\Paper2\XRD-Fluorescence.opj
Inte
nsity [cts
]
Diffraction Angle [°2]
T0
T05
T10
T25
T50
T75
CuK radiation, Ni-filternot stacked
10 20 30 40 50 60 70 80
0
500
1000
1500
2000
G:\Auftr_Proj\S-Auftraege\2012\S12_0008 XRD Course Riga\Paper Andris\Paper2\XRD-Fluorescence.opj
Inte
nsity [cts
]
Diffraction Angle [°2]
T0
T05
T10
T25
T50
T75
CuK radiation, Ni-filternot stacked
10 20 30 40 50 60 70 80
0
500
1000
1500
2000
G:\Auftr_Proj\S-Auftraege\2012\S12_0008 XRD Course Riga\Paper Andris\Paper2\XRD-Fluorescence.opj
Inte
nsity [cts
]
Diffraction Angle [°2]
T0
T05
T10
T25
T50
T75
CuK radiation, Ni-filternot stacked
Cu radiation, Ni filter in 2nd beam
(PANalytical X’Pert Pro)
100% TiO2
95% TiO2
+ 5% NiFe2O4
90% TiO2
+ 10% NiFe2O4
75% TiO2
+ 25% NiFe2O4
50% TiO2
+ 50% NiFe2O4
25% TiO2
+ 75% NiFe2O4

Secondary-monochromated Cu-Radiation
39
10 20 30 40 50 60 70 80
0
200
400
600
800
1000
1200
not stackedCuK radiation, Graphite monochromator
Inte
nsity [cts
]
Diffraction Angle [°2]
T0
T05
T10
T25
T50
T75
T100
G:\Auftr_Proj\S-Auftraege\2012\S12_0008 XRD Course Riga\Paper Andris\Paper2\XRD-Fluorescence.opj
10 20 30 40 50 60 70 80
0
200
400
600
800
1000
1200
not stackedCuK radiation, Graphite monochromator
Inte
nsity [cts
]
Diffraction Angle [°2]
T0
T05
T10
T25
T50
T75
T100
G:\Auftr_Proj\S-Auftraege\2012\S12_0008 XRD Course Riga\Paper Andris\Paper2\XRD-Fluorescence.opj
10 20 30 40 50 60 70 80
0
200
400
600
800
1000
1200
not stackedCuK radiation, Graphite monochromator
Inte
nsity [cts
]
Diffraction Angle [°2]
T0
T05
T10
T25
T50
T75
T100
G:\Auftr_Proj\S-Auftraege\2012\S12_0008 XRD Course Riga\Paper Andris\Paper2\XRD-Fluorescence.opj
10 20 30 40 50 60 70 80
0
200
400
600
800
1000
1200
not stackedCuK radiation, Graphite monochromator
Inte
nsity [cts
]
Diffraction Angle [°2]
T0
T05
T10
T25
T50
T75
T100
G:\Auftr_Proj\S-Auftraege\2012\S12_0008 XRD Course Riga\Paper Andris\Paper2\XRD-Fluorescence.opj
10 20 30 40 50 60 70 80
0
200
400
600
800
1000
1200
not stackedCuK radiation, Graphite monochromator
Inte
nsity [cts
]
Diffraction Angle [°2]
T0
T05
T10
T25
T50
T75
T100
G:\Auftr_Proj\S-Auftraege\2012\S12_0008 XRD Course Riga\Paper Andris\Paper2\XRD-Fluorescence.opj
10 20 30 40 50 60 70 80
0
200
400
600
800
1000
1200
not stackedCuK radiation, Graphite monochromator
Inte
nsity [cts
]
Diffraction Angle [°2]
T0
T05
T10
T25
T50
T75
T100
G:\Auftr_Proj\S-Auftraege\2012\S12_0008 XRD Course Riga\Paper Andris\Paper2\XRD-Fluorescence.opj
TiO2 + NiFe2O4,
Cu radiation, 2nd graphite monochromator
(PANalytical CubiX3)
95% TiO2
+ 5% NiFe2O4
90% TiO2
+ 10% NiFe2O4
75% TiO2
+ 25% NiFe2O4
50% TiO2
+ 50% NiFe2O4
25% TiO2
+ 75% NiFe2O4
100% NiFe2O4
In case of Fluorescence, use:
- Secondary-beam monochromator
- Energy-dispersive Detector
- Different X-ray tube (Cr / Fe / Co)

Summary: Monochromators
40
Optical Element Effect on Spectrum Effect on Intensity
Kβ Filter Reduces Kβ peaks Moderate loss
Graphite Monochromator Eliminates Kβ peaks Eliminates Fluorescence
Strong loss
Multi-bounce Monochromator Eliminates Kβ and Kα2 Eliminates Fluorescence
Massive loss (mostly used on Synchrotrons)
Energy dispersive Detector Reduces Kβ peaks Eliminates Fluorescence
No loss

Overview of Instruments
41
Lab Instrument Monochromator
RMS Foundation Bruker D8 Energy dispersive Detector
Uni Bern Panalytical X’Pert Kβ-Filter
Uni Bern Panalytical CubiX Graphite Monochromator
Bruker D8 Panalytical X’Pert Panalytical CubiX