Aldol condensation
-
date post
22-Oct-2014 -
Category
Education
-
view
369 -
download
4
description
Transcript of Aldol condensation

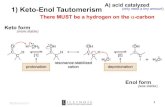
ALDOL CONDENSATION

Condensation between two molecules of an Aldehyde or a ketone to form a β-hydroxyaldehyde or a β-hydroxy ketone is known as a ALDOL CONDENSATION.
ALDOL CONDENSATION is possible only when the carbonyl compound contains atleast one
α-hydrogen atom.

Thus the following Aldehydes or ketones having no α-hydrogen atom do not undergo Aldol Condensation.

Some conditions regarding the
Aldol ReactionA reversible equilibrium
OH is the base typically used in an aldol reaction.
Aldol reactions can be carried out with either
aldehydes or ketones.
With aldehydes, the equilibrium favors products
With ketones the equilibrium favors the starting
materials.

MECHANISM OF ALDOL
CONDENSATION

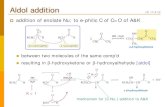
Step 1: First, an acid-base reaction. Hydroxide functions as a base and removes the acidic a-hydrogen giving the reactive enolate. Step 2: The nucleophilic enolate attacks the aldehyde at the electrophilic carbonyl C in a nucleophilic addition type process giving an intermediate alkoxide. Step 3: An acid-base reaction. The alkoxide deprotonates a water molecule creating hydroxide and the β-hydroxyaldehydes or aldol product.

Step:1
An enolate ion is the anion formed when an alpha hydrogen in the molecule of an aldehyde or a ketone is removed as a hydrogen ion.
enolate ion

Step:2
The alkoxide ion is the conjugate base of alcohols.

Alkoxide ion is protonated by water.
Step:3
aldol product

GENERAL FEATURES OF
ALDOL CONDENSATION

The aldol condensation is facilitated by-I groups on the carbonyl component and retarted by +I groups as described in CHзCHO and CHзCOCHз condensations.
The condensation is readily reversible and the position of equilibrium is not always favorable to the product.
Eg:

LIMITATIONS: Aldol products,as such, are not always
isolated from the reaction mixture.eg:acetaldol readily forms a cyclic hemi-acetol.
EXTENSIONS: Aldol condensation can occur between Two identical or different aldehydes Two identical or different ketones An aldehyde and a ketone

The R groups may be H, alkyl, or aryl. When the R
groups in one molecule are different than those in the
other, the reaction is called a crossed-aldol
reaction. The ability to join different aldehydes and
ketones together is what give this process its synthetic
value.
The name aldol condensation is also commonly
used,especially in biochemistry,to refer to just the first
stage of the process the aldol reaction itself as
catalysed by aldolases.

In its usual form ,it involves the nucleophilic
addition of a ketone enolate to an aldehyde
to form a β-hydroxy ketone or
aldol(aldehyde+alchohol), a structural unit
found in many naturally occuring molecules
and pharmaceuticals.

THANK YOU
Presented by -Akili Karuna









![Index [application.wiley-vch.de] · 1388 Index aldol condensation 477 – ultrasonic conditions 602 aldol cyclization 484 ... – Michael–aldol–dehydration 64 – Mukaiyama 247,](https://static.fdocument.org/doc/165x107/5f07e4047e708231d41f4542/index-1388-index-aldol-condensation-477-a-ultrasonic-conditions-602-aldol.jpg)

![α -Fluoro Aldehydes: Competition between Felkin Anh and ... · aldehyde carbon atom (Scheme 3). This feature,[1a, 5,9, 10] often present in aldol transition states involving (Z)-enolates,](https://static.fdocument.org/doc/165x107/5e1fdd1f732f4767d86521f6/-fluoro-aldehydes-competition-between-felkin-anh-and-aldehyde-carbon-atom.jpg)