ACMT Board Review 2012: Molecular Mechanisms1.3 Apoptosis: Programmed Cell Death Homeostatic...
Transcript of ACMT Board Review 2012: Molecular Mechanisms1.3 Apoptosis: Programmed Cell Death Homeostatic...
ACMT Board Review 2012: Molecular Mechanisms
Russ Kerns, MD, FACMT Carolinas Medical Center
Charlotte, NC
Objectives: Cover Core Content
1.2 Molecular components/mechanisms 1.2.1 Glycolysis & oxidative phosphorylation 1.2.2 Other metabolic pathways (β-oxidation) 1.2.4 Transport proteins (hemoglobin) 1.2.5 Channels
1.3 Cytotoxic mechanisms Provide key example toxins
Oxidative Phosphorylation
Energy is released when ATP → P + ADP Restoration of ATP (energy stores) involves
phosphorylation of ADP via coupling of oxidation of H+ to form H20.
Oxidative Phosphorylation
carbohydrate/fatty acid
acetyl-CoA
TCA
NADH/FADH2
electron transport chain
H+ + e-
P
ADP ATP H2O ADP
antiporter
H+
e- + O2- + H+
ATP synthase
H+ H+
Main carb metab path Cytosolic process 6C cmpd → 2 X 3 C cmpd Net 2 ATP molecules Make pyruvate for Kreb’s
Glycolysis glucose
glucose-6-P
fructose-6-P
glyceraldehyde-3-P + DHA-P
2 [pyruvate]
fructose-1,6-diP
2 [glyceraldehyde-3-P]
2 [1,3-diphosphoglycerate]
2 [P intermediates]
ATP
ATP
ATP
ATP
Arsenic (V) Substitutes for P Fail to make 1,3-diP intermed Fail to make ATP Fail to make pyruvate
Glycolysis: Toxins
glyceraldehyde-3-P + DHA-P
2 [glyceraldehyde-3-P]
2 [1,3-diphosphoglycerate]
2 [P intermediates]
ATP
2NAD+ + 2 P As5+ X
Glycolysis
CH3CO-CoA acetyl-CoA
NADH
CH3COCOOH
CoA, NAD+
CH3CH2OCOOH
NAD+
lactate
pyruvate
NADH, CO2
Kreb’s TCA Cycle pyruvate acetyl-CoA
NADH NAD+
citrate
Succinyl-CoA
oxalosuccinate
isocitrate
oxaloacetate
succinate
maleate cis-aconitate
α-ketoglutarate
fumarate
NAD+
NADH
NAD+ NADH
FADH FADH2
Kreb’s TCA Cycle: Toxins
Rodenticides Sodium monofluoroacetate Fluoroacetamide
acetyl-CoA
fluorocitrate
isocitrate
oxaloacetate
cis-aconitate monofluoroacetate
fluoroacetamide
Electron Transport Chain Mitochondrial process Series of oxidation-reduction reactions
Cytochrome enzymes FADH2 and NADH electron sources Produce H2O and ATP
Cytochrome Oxidase Inhibitors
acetyl CoA
lactate
glucose
pyruvate
TCA cycle
ATP H+
e- transport X X X
lactate
lactate
lactate lactate
lactate lactate
lactate
ETC: Toxins Uncouplers
Salicylate Dinitrophenol (explosives & wood preservative) Pentachlorophenol (fungicide)
Cytochrome aa3 inhibitors Cyanide H2S CO Methanol Phosphine gas Sodium azide (propellant in airbags)
NADH-CoQ reductase Rotenone (plant derived fish poison)
1.2.2 β-Oxidation of FFA
fatty acyl-CoA
CoA carnitine
fatty acylcarnitine fatty acyl-CoA
carnitine-palmitoyltransferase
CoA
(CH)nCO-CoA (CH)n-2CO-CoA + CH3CO-CoA acetyl-CoA
NADH, FADH2
CoA
(CH)nCOOH (CH)nCO-CoA acyl-CoA synthetase
ATP
fatty acyl-CoA
ADP + P CoA
β-Oxidation of FFA: Toxins
fatty acyl-CoA
CoA carnitine
fatty acylcarnitine fatty acyl-CoA
CoA
(CH)nCO-CoA (CH)n-2CO-CoA + CH3CO-CoA acetyl-CoA
NADH, FADH2
CoA
etoh, hypoglycin
valproate
β-Oxidation of FFA: Toxins ↑ NADH/NAD+ ratio
ethanol Hypoglycin(?)
Carnitine Valproate
Undefined mechanism Aflatoxin Amiodarone cereulide dimethylformamide tetracycline
www.australianprescriber.com
Mitochondrial DNA: NRTIs
Nucleoside reverse transcriptase inhibitors Mechanism
Inhibit mitochondrial DNA replication Inhibit ADP/ATP antiporter(?)
Result Lactic acidosis ± steatosis
Agents Stavudine Didandosine Zalcitobine Zidovudine Tenofovir (nucleotide)
X
Hemoglobin iron-based tetrameric protein α- and β-globin chains (2 each) Heme complex in each chain (4 total)
protoporhyrin ring central iron atom
Hemoglobin: Toxins
Site of action of toxins Heme synthesis Erythropoiesis Hemorrhage Oxidant stress Competition for oxygen binding
Heme Synthesis: Indirect Toxins
Acute Intermittent Porphyria Hepatic Autosomal dominant Reduced HMB synthase activity Some drugs may exacerbate AIP by
increasing ALA-synthase activity Poorly defined mechanism
Heme Synthesis: AIP
Barbiturates Carisoprodol Danazol Ethchlorvinyl Meprobamate Primidone Pyrazolones Trimethadione
dark red urine
www.porphyriafoundation.com
Hemoglobin: Toxins
Erythropoiesis Nephrotoxins - ↓ erythropoietin Pure rbc aplasia – rare
INH Hypoglycemics (chlorpropamide, tolbutamide) Phenytoin Sulfasalazine Valproate (single case report)
Hemoglobin: Aplastic Anemia
Immune mediated T lymphocytes release cytokines
Suppress hematopoietic stem cells Apoptosis (↑ Fas receptors on stem cells)
TNF
interferon-γ
Hemoglobin: Aplastic Anemia
DNA injury Direct DNA injury
Ionizing radiation Inhibition of DNA replication
Folate inhibitors (methotrexate) Intermediary metabolite that binds DNA
Benzene (quinone + free radicals) Tubulin inhibition during cell replication
Antimitotics (colchicine, vincristine, vinblastin)
metaphase arrest
metaphase.wordpress.com
Hemoglobin: Aplastic Anemia Antibiotics
Chloramphenicol Anti-convulsants
Carbamazepine, phenytoin Anti-inflammatory agents
Diclofenac, D-penicilamine, gold salts, indomethicin, phenylbutazone
Anti-neoplastic agents Alkylating agents (nitrogen mustards) Antibiotics (danorubicin, adriamycin) Antimitotics (colchicine, vinblastin, vincristine) Antimetabolites (purine and pyrimidine analogues)
Antipsychotics Chlorpromazine, clozapine
Hemoglobin: Aplastic Anemia Chemicals
Benzene, lindane Metals
Arsenic Miscellaneous
Acetazolamide, captopril, cimetidine, chlorpromazine, dapsone, fluoxetine, meprobamate, nifedipine, PTU, ticlopidine, tocainide
Radiation
Hemoglobin: Toxins
Megaloblastic anemia ↓ Vit B12 absorption
Colchicine, metformin, neomycin ↓ folate absorption
Etoh Impaired dihydrofolate reductase
Methotrexate Pyrimethamine Pyridium Trimethoprim
Hemoglobin: Oxidant Stress
Heme: deoxyhgb → Methgb (Fe2+→Fe3+) Prophyrin ring by sulfur: Sulfhgb Globin: Heinz body hemolytic anemia
Hemoglobin: Oxidant Stress
Protection from oxidant stress Ascorbic acid (Vit C) Glutathione (intact HMP, G6PD) Enzymatic
NADH-dependent reductase (Cytochrome b5 reduc) NADPH-dependent reductase (intact HMP, G6PD) Catalase Hydrogen peroxidase
Hemoglobin: Methemoglobin
Fe2+ state: deoxyhemoglobin carries oxygen Fe3+ state: methemoglobin results from
oxidation and does not carry hemoglobin
HgbFe2+ + O2 → HgbFe3+O2 → HgbFe2+ + O2 > HgbFe3+ + O2-
Hemoglobin: Methemoglobin
Cyt b5 or NADH dependent reductase NADPH-dependent reductase
HgbFe2+
HgbFe3+
cyt b5
cyt b5+ NADH
NAD+
Cyt b5 reductase
MB+
LMB
NADPH
NADP+
NADPH- dep reductase
glycolysis hexose monophosphate shunt
Hemoglobin: Methemoglobin
Chemicals aniline arsine chlorates (old strike matches) chlorobenzene copper sulfate napthalene nitrites (food contaminants & poppers) nitrates (food & well water) NOx (oxides of nitrogen) phenol
aniline
chloro- & nitrobenzene
phenol
Hemoglobin: Methemoglobin
Medications -caines (benzo-, lido-, prilo-) dapsone (sulfonamide deriv) methylene blue nitrites, nitrates phenacetin phenazopyridine -quines (chloro-, prima-) sulfonamide antibiotics
aniline
sulfanilamide
phenacetin
benzocaine
Hemoglobin: Sulfhemoglobin
Characteristics Same agents that induce methgb Not reversible Shifts O2-hgb dissociation curve to right H2S arguable, probably does not cause
sulfhgb, and hopefully would not be a great test item.
Hemoglobin: Hemolysis Mechanism
Oxidation of the globin chain Glutathione depletion or membrane injury
Characteristics Extravascular: mild forms Intravascular: severe forms
Anemia Free hgb (in serum and urine) Reticulocytosis Decreased haptoglobin
ww
w.re
side
nts.
path
olog
y.pi
tt.ed
u
Hemoglobin: Hemolysis
Hemolysis > methemoglobin Arsine (AsH3) Stibine (SbH3) Napthalene Copper sulfate
Hemoglobin: Non-oxidant, Immune-mediated Hemolysis
Type I (IgG-mediated) penicillin
Type IV (Cell-mediated) α-methyldopa
Na+ Channel Structure
Found in neurons, glial cells & myocytes 9 subtypes Tetrameric protein Transmembrane SCN gene
SCN5 – Brugada Syndrome Voltage-gated (myocardial) Ligand-gated (nicotinic)
Na+ Channel Modulation
Agonists – channel openers Aconitine (Monk’s Hood) Batrachotoxin (Poison Dart Frog) Ciguatoxin Grayanotoxin (Azalea & Rhododendron) Veratridine (Hellebore sp)
Na+ Channel Modulation
Antagonists Ia Antidysrhythmics
Procainamide, quinidine, disopyramide Ib Antidysrhythmics
Lidocaine, phenytoin Ic Antidysrhythmics
Encainide, flecainide, propafenone Others
Amiodarone, carbamazepine, cocaine, diphenhydramine, propranolol, propoxyphene, thioridazine and metabolite
Na+ Channel Modulation: Rate-Dependent baseline
7:50 min
2:50 min
QRS – 140 ms; BP - 145/78 mmHg
5:50 min
QRS – 160 ms; BP - 151/68 mmHg
QRS – 220 ms; BP - 0 mmHg QRS – 180 ms; BP -164/65 mmHg
Na Channel Recovery
Class Ia: τrecovery 1-10 sec Class Ib: τrecovery < 1 sec Class Ic: τrecovery > 10 sec
L-type Ca2+ Channel
Four proteins Span cell membranes Regulates calcium entry
Closed in resting state Require activation to open
Channel location determines the functional result of calcium entry
Ca2+ Channel Activation - Myocardial
Ca2+- mediated Ca2+ - release Result
↑HR ↑contractility
Modulators Catecholamines G protein cAMP protein kinase
Ca2+ Channel Activation - Vascular
Result vasoconstriction Maintenance of BP
Modulators α1 stimulation β2 stimulation angiotensin endothelin
Ca2+ Channel Antagonism
CCB drugs Nifedipine (dihydropyridine) Diltiazem (benzothiazepine) Verapamil (phenylalkylamine) Bepridil (diarylaminopropylamine)
Cyclic antidepressants Propafenone
Ca2+ Channel Agonist
Levosimendan Directly opens Ca2+ channel Heart failure treatment Experimental treatment of CCB toxicity No human overdose
K+ Channel Structure Tetrameric protein in the cell membrane
Central pore through which K+ flows
Normally closed Opening leads to K+ efflux from the cell
K+ Channel Function Inhibition of cell function
Acts to prevent overuse of the cell Opening stimuli
↓intracellular energy molecules (ATP) ↑intracellular Na+
↑intracellular Ca2+
ATP-K+ Channel Modulation
ins
ins
ins
ins ins
ins
K+ channel
Ca2+ Ca2+
Ca2+
Sulfonylurea (glipizide, glyburide)
ins ins
ins
Normal Function: Myocardial K+ Channel
Effective Refractory Period Depolarization not possible
Relative Refractory Period Depolarization possible with sufficient
electrical stimulus
RRP ERP
K+ Channel Modulation
K+ channel inhibition (Class III drugs) Prolongs action potential (phase 3) Equalizes refractoriness of ischemic and non-
ischemic tissues
X Na+!
Prolonged QTc / TdP Antidysrhythmic
Class I (quinidine and quinine) Class III (amiodarone, bretylium, dofetilide, ibutilide)
Antidepressants Serotonin agonists
Antihistamine terfenadine, astemizole
Antipsychotic haloperidol - butyrophenone thioridazine - phenothiazine sertindole - atypical
Prolonged QTc / TdP GI agents
cisapride Metabolic
hypokalemia (diuretics) hypomagnesemia (diuretics)
Metals arsenic
1.3 Antimitotics
Mitosis Forming of identical daughter cells by replicating and
dividing the chromosomes Replication occurs centrally in the parent cytoplasm Spindle apparatus attach to the chromosomes
(metaphase) and pull them towards the centromere (anaphase) prior to completion of cell division
www.cancerquest.emory.edu
Antimitotics
Spindle apparatus is composed of tubulin polymerized subunits polymerization ↔ depolymerization
Antimitotics interfere with spindle function Inhibit polymerization
colchicine, vincristine, vinblastine (vinca alkaloids) Inhibit depolymerization
taxol (alkaloid from Yew) Result: metaphase arrest
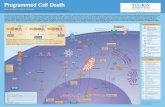
1.3 Apoptosis: Programmed Cell Death
Homeostatic mechanism for removal of: damaged, infected, aged cells activated immune cells (no longer needed)
Non-inflammatory Extrinsic triggers Intrinsic triggers
Apoptosis
Results Caspase family activation (3,7,8,9,10)
Cleaves DNA, protein Apoptotic protein activation (BH3 family)
Initiates mitochondrial pore formation
Apoptosis: Programmed Cell Death
Progressive condensation of nuclear contents Nucleus lyses (karyorhexis) Cell shrinkage, cytoplasmic condensation Apoptotic bodies, “budding” formed Macrophages remove apoptotic bodies
Normal lymphocyte
apoptotic lymphocyte